Microalgae Bioreactors Explained: Producing Phycocyanin, Astaxanthin, and High-Value Compounds Off-Grid
Microalgae bioreactors produce phycocyanin and astaxanthin in closed-loop systems, balancing efficiency, yield, and off-grid resilience.
Microalgae as Survival Infrastructure: Phycocyanin, Astaxanthin, and the Future of Decentralized Bioproduction
Summary
Microalgae-based systems represent one of the most compact and versatile forms of biological production currently available. Unlike traditional agriculture or industrial chemical synthesis, microalgae can operate within closed-loop environments, producing high-value compounds such as phycocyanin and astaxanthin with minimal land use and tightly controlled resource inputs.
In the context of survivalism, decentralized infrastructure, and speculative futurism, microalgae should not be viewed solely as a nutritional supplement source, but as a modular bioreactor platform. This has the potential to produce not only food, but also pigments, antioxidants, and specialized biochemical outputs under constrained conditions.
At the same time, the processes that enable high-purity compound extraction introduce dependencies on industrial infrastructure, chemical inputs, and technical expertise. This creates a fundamental tradeoff between resilience and efficiency, which becomes especially relevant under conditions of supply chain disruption or off-grid operation. Phycocyanin and astaxanthin provide a useful lens through which to examine these dynamics.
1. Microalgae as a Bioreactor Platform
Microalgae systems function as living chemical factories, converting light, carbon dioxide, water, and nutrients into structured biological compounds. Unlike conventional crops, which prioritize caloric output, these organisms can be cultivated for highly specific functional outputs, including proteins, pigments, and bioactive molecules.
What makes microalgae particularly compelling is their ability to operate within controlled environments. They can be grown in enclosed photobioreactors or integrated into biodome systems where light, temperature, and nutrient flow are managed with precision. This allows for high productivity per unit area while reducing exposure to environmental variability.
Microalgae blur the line between agriculture and manufacturing. They are not simply “grown” in the traditional sense; they are operated as production systems, capable of generating targeted outputs under defined conditions. This positions them as a foundational technology for decentralized and closed-loop environments.
2. Phycocyanin and Astaxanthin as Synthesis Products
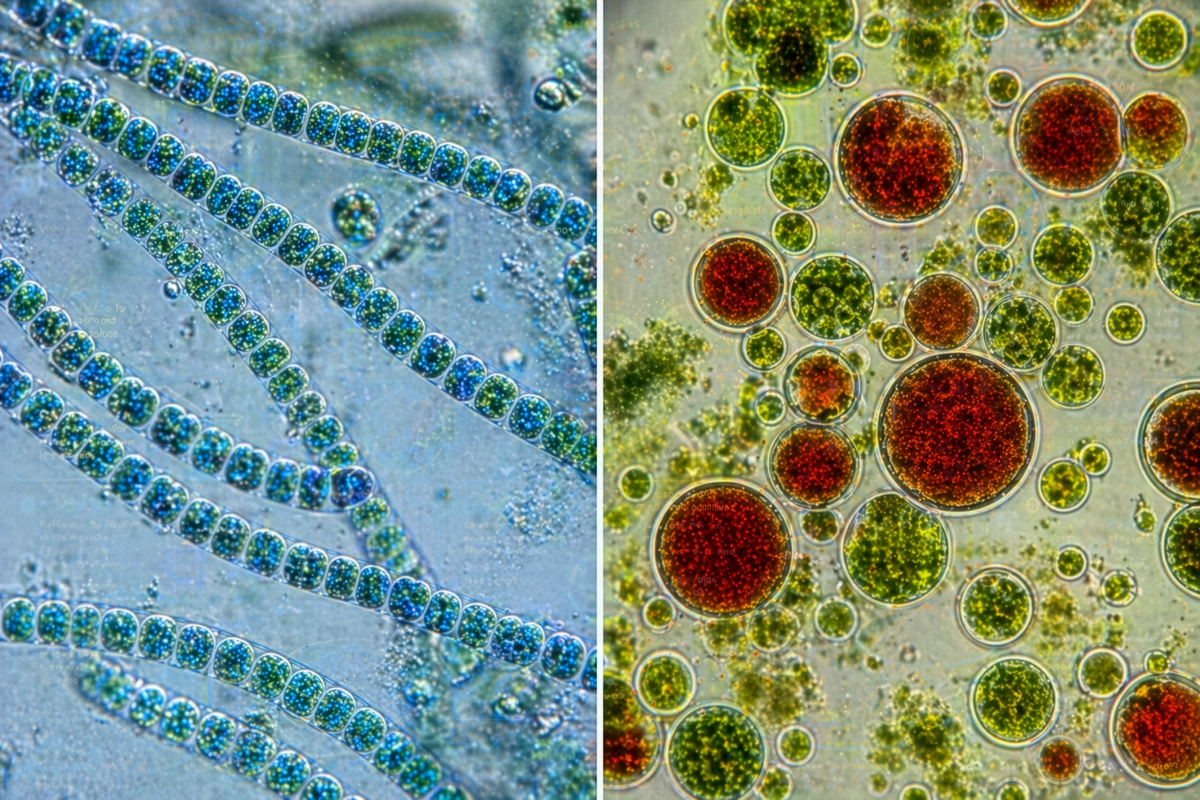
Phycocyanin, derived from spirulina, is a blue pigment-protein complex widely used in food, nutraceuticals, and research applications. Its value is tied not only to its function as a natural colorant, but also to its antioxidant and anti-inflammatory properties. Spirulina itself is relatively resilient and can be cultivated in moderately controlled environments, making phycocyanin one of the more accessible microalgae-derived compounds.
Astaxanthin, by contrast, represents a more complex production pathway. Derived primarily from the microalga Haematococcus pluvialis, it is a red carotenoid known for its extremely high antioxidant capacity. It is used extensively in aquaculture, supplements, and cosmetics. However, its production typically involves stress-induced growth phases and more tightly controlled environmental conditions, making it both higher in value and more demanding to produce.
Together, these two compounds illustrate the range of what microalgae systems can produce, from relatively accessible outputs to highly specialized, high-value biochemical products.
2.1 Practical Applications of Spirulina and Astaxanthin
Spirulina-derived products, particularly phycocyanin, serve multiple roles across nutrition, industry, and basic biochemical use. As a high-protein biomass, spirulina can function as a direct food supplement, while its pigment compounds are used as natural colorants in food and cosmetic formulations. Beyond aesthetics, phycocyanin has applications as an antioxidant compound and as a reagent in certain analytical and research contexts. In contrast, astaxanthin is a higher-value carotenoid primarily utilized for its strong antioxidant properties. It is widely used in aquaculture to enhance coloration in fish and crustaceans, as well as in human supplements. Together, these compounds represent a spectrum of outputs ranging from bulk nutritional support to concentrated, high-value biochemical products that can be stored, traded, or integrated into more specialized production chains.
3. Production Systems and the Resilience Tradeoff
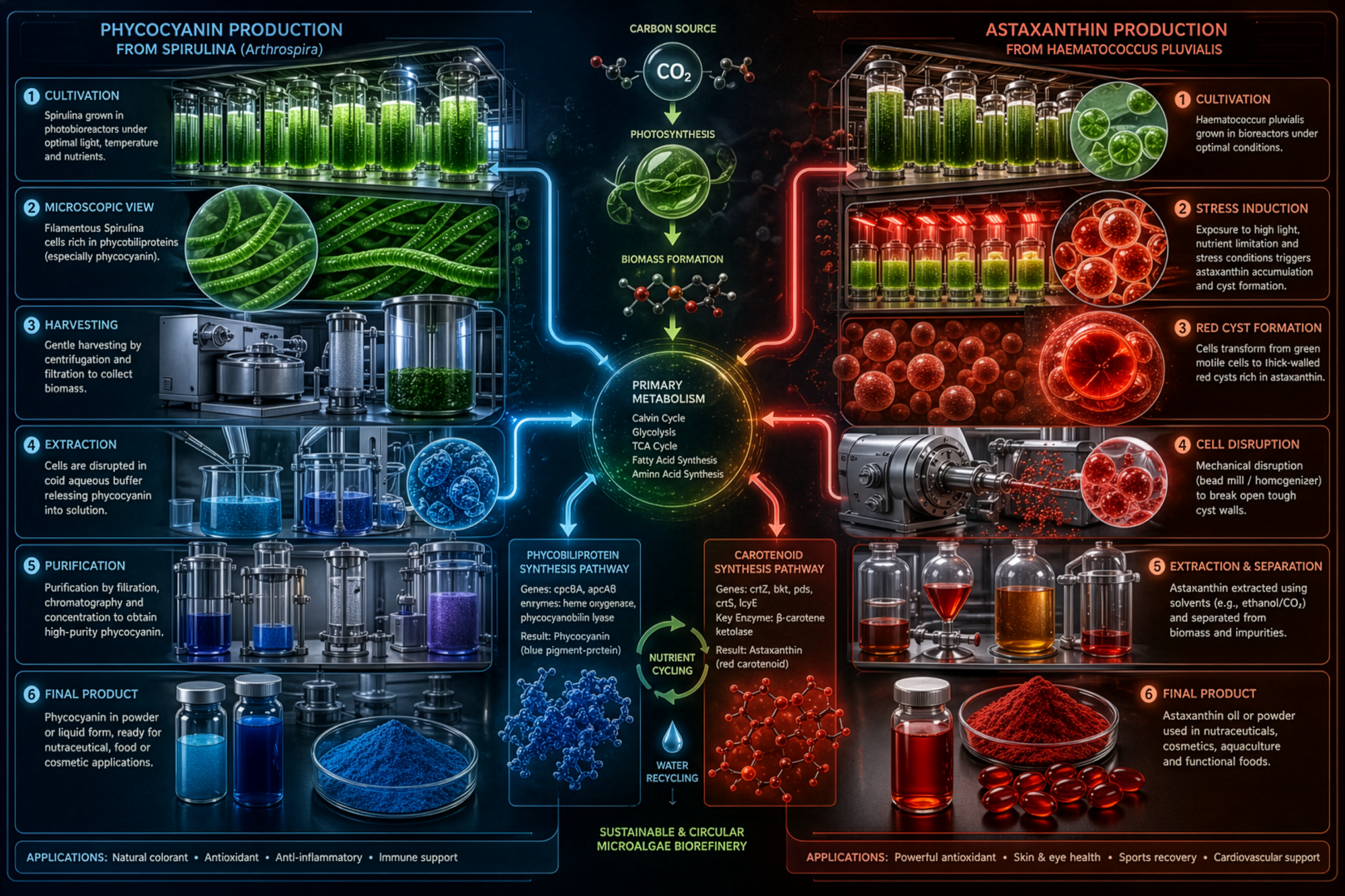
The production of microalgae-derived compounds generally follows a consistent pipeline, beginning with cultivation and progressing through harvesting, cell disruption, and extraction. While the stages are conceptually straightforward, the complexity of each step varies significantly depending on the desired output.
At one end of the spectrum are lower-complexity systems that prioritize resilience. These systems are easier to deploy, rely on fewer specialized inputs, and are more adaptable to off-grid or resource-constrained environments. However, they tend to produce lower yields and less refined outputs.
At the other end are high-complexity systems designed for efficiency. These systems use advanced photobioreactors, tightly controlled environmental conditions, and multi-stage processing pipelines to achieve high purity and yield. The tradeoff is that they require significant capital investment, technical expertise, and access to specialized infrastructure.
This creates a fundamental tension. Systems optimized for independence and resilience tend to sacrifice efficiency, while systems optimized for performance tend to become dependent on broader industrial support.
Microalgae Bioreactors (2026): Phycocyanin, Astaxanthin, and Decentralized Bioproduction Explained
4. Extraction as the Critical Bottleneck
One of the most important constraints in microalgae-based production is extraction. Growing biomass is relatively straightforward compared to isolating and refining specific compounds from within that biomass.
High-efficiency extraction methods often depend on controlled industrial processes and specialized inputs. This introduces a layer of dependency that is easy to overlook when focusing only on cultivation. The ability to grow microalgae locally does not automatically translate into the ability to produce high-purity outputs locally.
In practical terms, this means that extraction becomes a structural bottleneck. It is the point at which decentralized systems intersect with centralized industrial capabilities. Without addressing this stage, microalgae systems remain partially dependent, even if cultivation is fully localized.
5. Supply Chain Fragility and Localized Production
Modern nutraceutical and biochemical supply chains rely on global logistics, specialized facilities, and consistent access to processing inputs. Under stable conditions, this allows for high efficiency and large-scale production. Under disruption, however, these systems can become fragile.
Microalgae-based systems offer a degree of insulation by localizing the initial stages of production. Biomass can be generated on-site, reducing dependence on agricultural supply chains. However, without local processing capabilities, the system remains incomplete.
This creates a layered model of resilience. Cultivation is the most accessible and robust layer, while processing introduces moderate dependency, and high-purity refinement remains the most infrastructure-intensive stage. Understanding this hierarchy is critical when evaluating the role of microalgae in survival-oriented or decentralized systems.
6. Integration into Closed-Loop Systems
Within closed-loop biodome environments, microalgae can perform multiple roles simultaneously. They contribute to oxygen production, carbon capture, and nutrient cycling while also generating useful biochemical outputs. This allows them to integrate naturally with other subsystems such as hydroponic agriculture and water recycling.
The value of microalgae in this context is in how they interact with the broader system. They act as both a production layer and a stabilizing component within a larger ecological network. This makes them particularly well suited for environments where efficiency, redundancy, and resource cycling are all critical.
7. Future Outlook: Decentralized Bioproduction
Looking forward, microalgae systems have the potential to evolve into modular bioreactor units that can be deployed in remote or resource-constrained environments. They may become standard components of off-grid infrastructure, integrated into biodomes, smart buildings, or experimental biomanufacturing systems.
The conceptual shift is significant. Rather than viewing algae as something to be cultivated passively, these systems can be understood as deployable biological infrastructure. They represent a step toward distributed production models where biochemical outputs are generated locally rather than sourced globally.

8. Applied Scenarios:
8.1 A Distributed Microalgae Production Barn
Consider a small, semi-autonomous community operating as part of a broader regional federation. It is not fully isolated, but it prioritizes local production capacity for critical inputs - nutritional, biochemical, and environmental. Rather than constructing a purpose-built laboratory, the community repurposes an existing agricultural structure: an aging pole barn.
The interior is divided into a series of controlled bays, each retrofitted to function as a contained microalgae production lane. The original livestock stalls become modular compartments, fitted with translucent panels, LED lighting arrays, and basic environmental controls. Water circulation, nutrient dosing, and aeration are centralized, but each bay operates semi-independently.
In practice, the barn does not function as a single “reactor,” but as a distributed set of bioreactors, each tuned to a specific role.
One section is dedicated to spirulina cultivation. These vats operate in relatively stable conditions, optimized for consistent biomass production. The output is periodically harvested, dried, and used directly as a nutritional supplement or processed further for pigment extraction. Because spirulina is comparatively robust, this lane acts as the baseline production layer; reliable, predictable, and relatively forgiving.
A separate section houses Haematococcus pluvialis, the organism used for astaxanthin production. Unlike spirulina, this process is staged. Initial growth occurs under favorable conditions to build biomass, followed by a controlled stress phase; light intensity, nutrient limitation, or other environmental shifts to trigger astaxanthin accumulation. These bays require more attention and tighter control, and their output cycles are less continuous but significantly higher in value.
Between these two primary lanes, additional bays are used for intermediate roles. Some are allocated for inoculation and culture scaling, ensuring that fresh, uncontaminated cultures can be introduced into production vats. Others are used for biomass concentration and preliminary processing, where harvested material is thickened, filtered, or prepared for downstream handling.
The barn’s infrastructure reflects a balance between simplicity and control. It is not a pharmaceutical facility, but it is not a passive agricultural space either. The system relies on:
- stable water inputs and recycling
- consistent power for lighting and circulation
- basic monitoring of temperature, pH, and growth conditions
Failures are localized. If one bay becomes contaminated or underperforms, it can be isolated without compromising the entire operation. This modularity mirrors the community’s broader design philosophy: avoid single points of failure, even within production.
The output of the barn feeds directly into the community’s internal economy. Spirulina-derived products contribute to baseline nutrition and trade within the federation, while astaxanthin (due to its higher value density) is reserved for specialized use or external exchange. The facility does not attempt to match industrial purity standards, but it produces enough functional output to reduce reliance on external supply chains.
Over time, the barn evolves. Lighting systems are upgraded, water loops become more efficient, and certain bays are reconfigured as the community’s needs change. What began as a retrofit becomes a living production asset, shaped by both biological constraints and human decision-making.
In this model, microalgae are no longer an abstract biotechnology concept. They become a tangible layer of local production and integrated into existing structures, scaled to community needs, embedded within a wider network of exchange.
8.2 Applied Scenario: High-Density Microalgae Production in a Closed-Loop Biodome
In a more advanced configuration, microalgae production is no longer retrofitted into existing structures but designed as an integrated component of a purpose-built biodome. Within a federation of semi-autonomous communities, one node specializes in high-density bioproduction, operating a controlled environment where biological output is treated with the same precision as energy or water systems.
The biodome is organized vertically, with microalgae photobioreactors embedded along interior walls and suspended in layered arrays. These are not open vats, but sealed, transparent tubing systems that maximize surface area exposure to controlled light sources. Spectrally tuned LED panels adjust wavelength and intensity in real time, optimizing growth conditions based on the metabolic phase of each culture. Spirulina lanes operate under stable, high-throughput conditions, continuously cycling biomass through growth and harvest phases, while adjacent Haematococcus lines shift dynamically between expansion and stress states to induce astaxanthin accumulation.
All inputs are tightly regulated. Carbon dioxide is captured from adjacent processes, either from human occupancy, fermentation systems, or small-scale combustion sources, fed directly into the algae loops. Water is continuously filtered, sterilized, and recirculated, with nutrient concentrations monitored and adjusted through automated dosing systems. Waste heat from other dome subsystems is reclaimed to maintain temperature stability, reducing overall energy demand.
Unlike the barn-based model, where processes are semi-manual and compartmentalized, the biodome operates as a synchronized biological network. Sensors track parameters such as pH, dissolved oxygen, turbidity, and growth rates, feeding data into a control layer that adjusts environmental conditions across the entire system. Rather than reacting to problems after they emerge, the system operates predictively, smoothing fluctuations before they impact output.
Processing is co-located but separated into controlled modules. Biomass is harvested through continuous-flow separation, then routed to enclosed processing units where cell disruption and compound isolation occur under standardized conditions. The goal is not full industrial purity, but a consistent and reliable intermediate-grade output that can be used internally or refined further within the broader network.
The role of the biodome within the federation is distinct. It is not designed for redundancy in the same way as smaller, distributed setups. Instead, it acts as a specialized production hub, trading higher-value compounds for raw inputs, energy, or other materials produced by partner communities. Where the barn model prioritizes independence, the biodome prioritizes efficiency, throughput, and integration.
Over time, the system evolves through iteration rather than expansion alone. New strains are introduced, lighting profiles are refined, and control algorithms improve yield consistency. The biodome becomes less of a static facility and more of a continuously tuned environment, where biological production is treated as an engineered process rather than a passive one.
In this configuration, microalgae are fully embedded into the architecture of advanced living environments. They are no longer an auxiliary subsystem, but a core layer of production, operating alongside energy, water, and food systems to form a tightly integrated, high-performance habitat.
Conclusion
Phycocyanin and astaxanthin highlight both the promise and the limitations of microalgae-based systems. They demonstrate that high-value biochemical production is possible within compact, controlled environments, but also reveal the dependencies that emerge when moving from raw biomass to refined output.
The central tradeoff remains clear. Systems designed for resilience emphasize accessibility and independence, while systems designed for efficiency emphasize precision and output at the cost of complexity and dependency.
In the context of survivalism, speculative futurism, and deep-tech system design, microalgae occupy a unique position. They are not simply a food source or a niche biotechnology, but a foundational component in the emerging architecture of decentralized biological production.