DIY Indoor Aeroponics: Efficiency, Energy Costs, and Scalable System Design
Indoor DIY aeroponics is water-efficient but highly sensitive; yields hinge on precision, while electricity (lighting) becomes the main constraint.

Indoor DIY Aeroponics for Personal Food Production
Summary
Indoor DIY aeroponics (growing plants with roots suspended in an enclosed chamber and intermittently wetted with a nutrient mist) can deliver high space-efficiency and very low direct water use, but it is system-sensitive: small errors in atomization quality, spray timing, water chemistry (pH/EC), sanitation, and indoor climate can rapidly degrade yields or cause crop loss.
The most decision-relevant tradeoff for personal food production is that indoor aeroponics often shifts the limiting factor from land/water to electricity (primarily lighting). This pattern is consistent across controlled-environment lettuce analyses: hydroponic controlled-environment agriculture can use ~15 kWh/kg lettuce supplied for lighting/cooling/ventilation/pumping, and associated footprints depend strongly on the electricity mix. [2] Comparable greenhouse-focused modeling for lettuce found far lower direct water use than field systems but substantially higher energy per kg, highlighting energy as the dominant sustainability constraint absent low-carbon power.
For DIY adoption, a pragmatic architecture is a modular “root-chamber + lighting bay” that can start as a 4-plant pilot and scale to 12+ plants by adding identical chambers and/or shelves. The engineering priority is reliability: robust filtration and anti-clog design, redundant timers/alarms, and a cleaning/sanitation regime oriented around biofilm prevention and “food-contact surface” thinking.
Assumptions where unspecified (explicitly used in the analytical models below): (i) personal (non-commercial) production, (ii) leafy greens/herbs as primary crops, (iii) full-indoor lighting (no meaningful sunlight contribution), (iv) a “good consumer LED” at ~2.5 µmol/J fixture efficacy as a planning value, and (v) electricity price scenarios of $0.15–$0.35/kWh to reflect large regional variance. Lighting assumptions are anchored to extension guidance on lettuce PPFD and DLI.
Definitions and System Types
Core definition
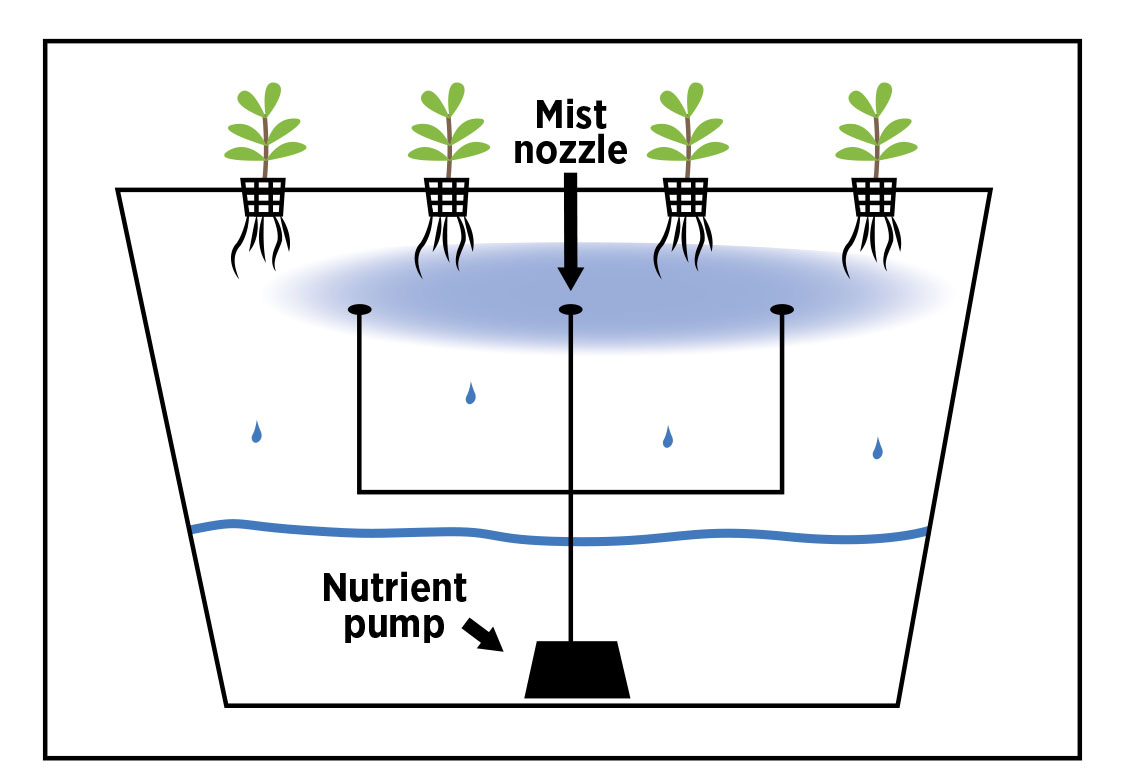
A practical, extension-style definition of aeroponics is: plant roots are suspended in an enclosed chamber and sprayed with a nutrient mist at short intervals (often seconds every few minutes), rather than being immersed or flowing in a film. [6]
A recent peer‑reviewed engineering paper frames aeroponics as periodic deposition of nutrient droplets onto hanging roots, with the hypothesized advantage of enhanced root aeration (oxygen availability) relative to some hydroponic root submersion configurations. [7] A complementary peer‑reviewed review in New Phytologist conceptualizes the aeroponic irrigation cycle as deposition → retention (thin-film formation) → decay (evaporation/gravity removal) and highlights strong coupling between aerosol physics, root architecture, environmental humidity, and nutrient solution properties. [8]
System taxonomy
Aeroponic DIY systems are best categorized by how droplets (or “fog”) are generated, because that choice drives pressure requirements, clogging risk, electrical complexity, maintenance burden, and yield sensitivity. [9]
| Type | Aerosol Generation Method | Practical Implication for DIY | Representative Evidence Anchors |
|---|---|---|---|
| Low-pressure aeroponics (LPA) | Pumped liquid through nozzles at relatively low pressure; often coarser spray | Lowest cost and simplest plumbing; higher sensitivity to uneven wetting and droplet coalescence; can be “good enough” for greens if spray timing is conservative | Atomization quality matters; droplet size affects oxygen availability and deposition dynamics [9] |
| High-pressure aeroponics (HPA) | Liquid supplied to atomizing nozzles under high pressure, focusing on stable atomization and coverage | Higher component rigor (rated tubing/fittings, filtration, pressure regulation); improves uniformity and reduces chamber dry spots when well-designed | Engineering design uses pressure atomizers and pressure tanks; droplet size targets often cited in the 30–100 micrometer range, with context-dependent nuance [10] |
| Fogponics | Ultrasonic atomizers generate very fine droplets (fog), often with more uniform distribution | Eliminates nozzle clogging but introduces new risks: water ingress into electronics, mineral scaling on transducers, and nutrient concentration drift due to evaporation | Peer-reviewed comparisons describe ultrasonic atomizers as liquid low-pressure standing systems with monodisperse droplet distributions [11] |
| Hybrid / staged systems | Combination of methods (e.g., pressure mist for fertigation plus airflow/humidity control, or coarse spray plus fog) | Used to manage different growth stages (seedling vs vegetative) and reduce risk of under-wetting during transitions | Literature emphasizes developmental-stage optimization and dosing regimen design across growth phases [12] |
Key nuance on “droplet size targets.” An engineering design paper notes that many sources cite an “ideal droplet diameter between 30–100 µm,” while also emphasizing that this figure traces to microgravity research and may not transfer directly without context; it further argues droplets much below ~1–2 µm may not deposit efficiently on roots unless velocities are very high (energy costly), while very large droplets can fall out rapidly. [13]
Evidence on yield sensitivity to atomization + timing
A controlled study on butterhead lettuce grown aeroponically (multiple atomizers with different droplet diameters and different spray intervals) found strong interactions between droplet size and spray interval for growth and yield proxies; in that experiment, the best-performing treatment reported shoot fresh weight ~65.74 g/plant, while poor atomizer/interval combinations produced much lower biomass. [14] This is less a “headline yield figure” than a risk signal: aeroponics can underperform hydroponics/soil if mist delivery is mis-specified. [15]
DIY Designs, Components, and Space Planning
Typical DIY Architectures
Most DIY systems converge to one of three physical layouts:
Enclosed root chamber + top plant deck (tote/bin format). Plants sit in net cups or collars in a lid; roots hang into a dark chamber where they are misted; solution drains back to a reservoir (same tote or separate). This matches extension descriptions of aeroponics as roots suspended in an enclosed chamber with misting on short cycles. [6]
Rack-mounted shelf modules (2×2 or 2×4 footprint). One or more chambers per shelf, each with its own lighting bay; scaling is done by adding shelves or duplicating modules. Lighting and heat management become more important in stacked systems because vertical clearance is constrained and heat can affect the shelf above. [16]
Vertical/tower variants (space-saving, higher airflow complexity). Towers are attractive in apartments but can create harder-to-control microclimates and nonuniform wetting unless engineered carefully; they also impose higher failure costs if a single pump outage affects many plants.
Core component stack (what “must work”)
A robust DIY aeroponic module can be decomposed into subsystems that mirror the engineering literature:
1) Nutrient mixing and storage. A dosing/mixing approach may use RO water and nutrient stock concentrates (common in engineered systems) and requires tracking pH and EC; extension guidance places typical nutrient-solution pH in ~5–6 for soilless culture. [17]
2) Atomization and distribution. Options include pressure atomizers, air-assist atomizers, or ultrasonic atomizers, each with distinct “requirements at the point of atomization” (high-pressure liquid vs low-pressure/standing + electricity). [18]
3) Control and automation. Aeroponics is timer-intensive; an extension fact sheet notes aeroponics typically requires a short-cycle timer running a pump for a few seconds every couple of minutes (illustrative magnitude). [6]
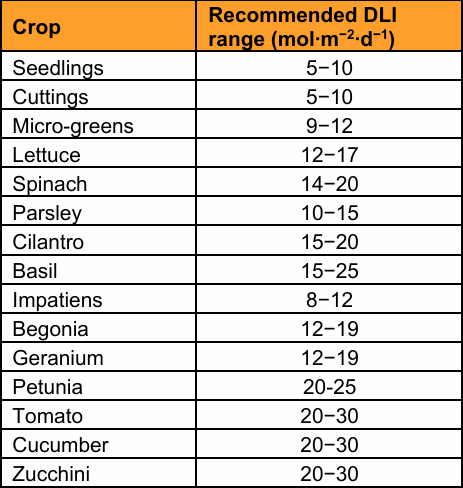
4) Lighting and canopy microclimate. Lettuce PPFD guidance for controlled environments is commonly ~250–350 µmol·m⁻²·s⁻¹; exceeding that can waste energy without meaningful yield gain and may risk photoinhibition. [16] Crop planning should also be DLI-driven; a Virginia extension publication lists lettuce DLI guidance of ~12–17 mol·m⁻²·d⁻¹. [19]
5) Monitoring and measurement. At minimum: nutrient solution pH and EC, temperature, and reservoir level; plant growth issues can be linked to pH management. A greenhouse/hydroponic guidance note emphasizes measuring and adjusting pH frequently and reports yield loss when pH management is neglected (lettuce sensitivity). [20]
Apartment vs house layout options
Apartments (high constraints): Primary risks are water leakage, noise, and humidity/mold. A recommended pattern is a single “contained” module (2×2 footprint) with secondary containment (spill tray), conservative reservoir volume, and ventilation that does not dump humid air into cold corners (condensation risk). Heat and clearance are nontrivial in stacked systems; extension guidance explicitly flags heat generation and vertical clearance as crucial in vertical/shelf systems. [16]
Houses (lower constraints): More options exist for dedicated grow rooms, larger reservoirs, and integrating ventilation. However, the same “single-point failures” apply: pump, timer, and sanitation. The research literature emphasizes that aeroponics adds cost/complexity for atomization control and reliability relative to simpler hydroponics, which is why engineered design decisions (pressure stability, filtration, sensorization) matter. [9]
Crop Suitability and Yield Expectations
Crop suitability: what is “aeroponics-friendly” for DIY
Most suitable (low structural burden, fast cycle): leafy greens (lettuce, arugula, kale) and many culinary herbs. Lettuce is routinely used in controlled-environment research and extension examples for DLI/PPFD planning. [21]
Moderately suitable (more training, higher light demand): basil and other vigorous herbs. A basil aeroponics study used a high-pressure irrigation system spraying roots for 25 seconds every 5 minutes, maintaining pH ~5.6 and EC ~1.70 dS/m, illustrating the level of control used in successful herb aeroponics. [22]
Challenging but feasible (high light + support + longer cycle): small fruiting crops (e.g., cherry tomato, peppers). A peer‑reviewed aeroponics review cites results where aeroponic yields for several crops exceeded soil culture (e.g., basil, parsley, cherry tomato, squash, bell pepper, red kale with reported percentage increases vs soil) and notes a study reporting greater tomato fruit mass in aeroponics vs hydroponics (1.95 g/fruit vs 1.56 g/fruit in that cited comparison). [23]
For home systems, the principal constraint for fruiting crops is usually light and space (DLI requirements). A Purdue extension note states tomatoes and strawberries can require >20 mol·m⁻²·d⁻¹ DLI; substantially above lettuce target, directly driving electricity use and heat in indoor setups. [24]
Yield expectations: framing for personal production
Aeroponic yields are best treated as a range of outcomes conditioned on design maturity, not a single number.
For butterhead lettuce, one controlled aeroponic study notes prior reports that lettuce yield could be ~50–150 g/plant (contextual literature range) and measured best-treatment shoot fresh weight ~65.74 g/plant under its tested atomization/interval regime. [25] This implies two operational takeaways for DIY: 1) Expect a commissioning period where yields are on the low end until spray timing, nozzle performance, and nutrient chemistry are stabilized. [15]
2) Yield is a control-system output. Atomizer and interval interactions can dominate outcomes, especially for small root chambers with uneven mist coverage. [26]
A useful “sanity-check” benchmark comes from greenhouse hydroponic modeling (not aeroponics but relevant to indoor soilless expectations): annual lettuce yields were modeled at ~41 ± 6.1 kg/m²/year for hydroponics versus ~3.9 ± 0.21 kg/m²/year for conventional field production in a specific regional comparison. [3] For indoor DIY aeroponics, hitting greenhouse hydroponic productivity levels requires strong lighting, climate management, and high uptime; conditions that are possible but not automatic.
Energy, Water, Nutrient, Cost, and Environmental Footprint Models
Lighting and energy fundamentals (dominant driver)
Crop light targets. Lettuce planning values commonly appear as:
· DLI: 12–17 mol·m⁻²·d⁻¹ (extension guidance). [19]
· PPFD: 250–350 µmol·m⁻²·s⁻¹ (controlled environment guidance). [16]
Lighting efficiency reality. Modern LED fixtures can exceed ~2.5 µmol/J (extension guidance), while peer‑reviewed LED efficacy analysis discusses physical/engineering limits and losses, including reductions when fixtures require protective design for high humidity/water exposure. [27]
System-level reality. Life-cycle analysis for hydroponic controlled-environment lettuce supply chains reports ~15 kWh electricity per kg lettuce supplied for lighting/cooling/ventilation/pumping (system-level, not just LEDs). [2] For DIY aeroponics, pump energy is typically minor relative to lighting; the high leverage is LED choice, photoperiod strategy, and managing heat/humidity so you do not need energy-intensive dehumidification or cooling. [28]
Example “1-, 4-, 12-plant” resource and cost models (illustrative)
These models are intentionally transparent and tunable. They assume leafy greens (lettuce-like) targets and a 16-hour photoperiod to meet DLI targets, consistent with common CEA practice and PPFD/DLI guidance. [29]
Assumption set (override as needed):
· Lighting:
· 1 plant: ~30 W average LED draw (small fixture or dimmed larger board).
· 4 plants: AC Infinity[30] IONBOARD S22 (115 W, 2×2 coverage) as an example reference. [31]
· 12 plants: AC Infinity[30] IONBOARD S24 (220 W, 2×4 coverage) as an example reference. [31]
· Fans/pumps (average power): 1–2 W average pump duty for misting; 10–20 W continuous for circulation/ventilation (varies widely by enclosure).
· Electricity price: scenario range $0.15–$0.35/kWh.
Illustrative annual electricity use
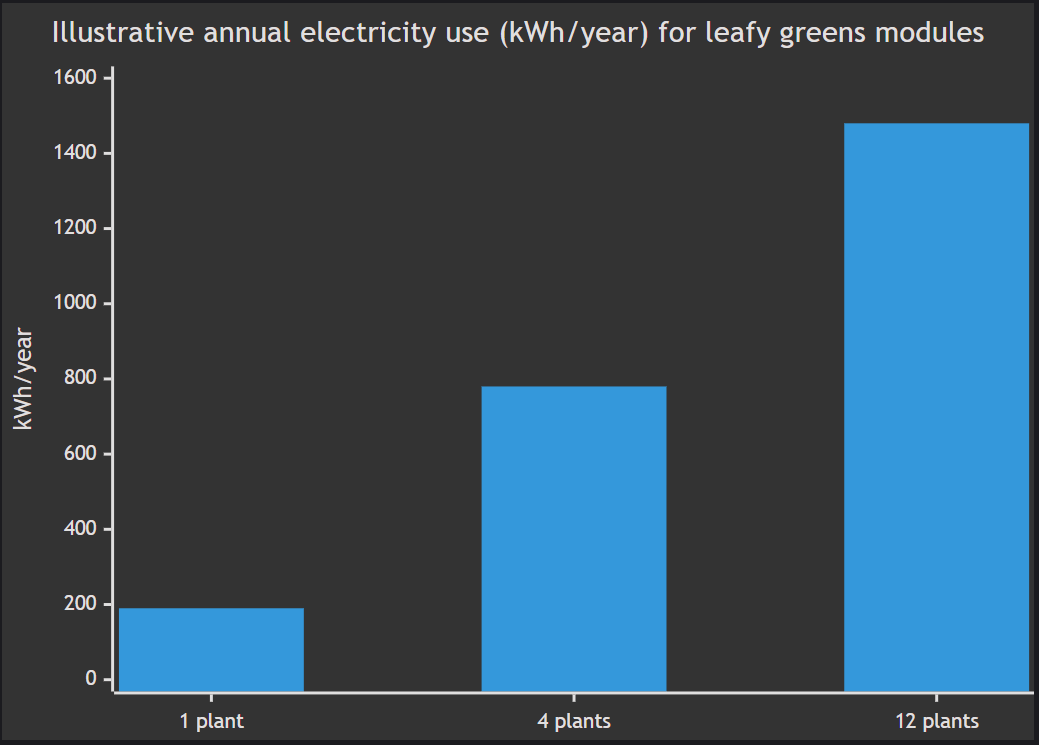
Interpretation: the 12-plant module is typically a 25–40 W/plant electricity commitment averaged over the year in a full-indoor scenario, dominated by lighting. This is directionally consistent with the broader LCA literature identifying electricity as the major hotspot for CEA lettuce under typical grid mixes. [32]
Direct water use (order-of-magnitude)
Closed soilless systems can use orders of magnitude less direct water than field systems, and a classic modeled comparison for lettuce reported hydroponic water demand around ~20 L/kg versus ~250 L/kg for conventional production (regional case). [33]
For personal modules, direct water consumption is largely plant transpiration plus small losses. As a planner’s heuristic (not a universal constant), using 20 L/kg produce for closed-loop soilless production provides an upper-bound style estimate when you do not yet have measurements. [3]
CAPEX/OPEX budgets (example ranges)
Budgets vary sharply with (i) whether you choose HPA vs LPA, (ii) whether you add “commercial-style” monitoring (pH/EC meters) and ventilation hardware, and (iii) local pricing. Product references below use manufacturer pages where available.
Capex budget ladder (USD, illustrative)
| System Size | “Basic Functional” Build | “Risk-Reduced” Build (Recommended) | Main Drivers |
|---|---|---|---|
| 1 plant | $80–$180 | $200–$450 | Lighting plus a reliable timer; secondary containment |
| 4 plants | $180–$400 | $450–$950 | Quality LED board; stable, cleanable atomization; pH and EC measurement |
| 12 plants | $350–$750 | $900–$1,800 | Stronger lighting (2x4); ventilation; higher-reliability atomization (often HPA features) |
Why the “risk‑reduced” column can dominate: aeroponics is sensitive to pH/EC drift and sanitation, and designs that treat water-contact surfaces as cleanable “food contact surfaces” require better access, filtration, and measurement tooling. [34]
Opex (monthly) for leafy greens (dominant = electricity):
· Electricity = (kWh/month) × (your $/kWh).
· Nutrients + pH adjusters + seeds: often secondary relative to electricity in full-indoor setups (scenario statement aligned with LCA hotspot findings). [32]
Sample bill of materials (BOM) with suppliers
Below is a “12-plant, 2×4 footprint” reference BOM designed for modular scaling. It includes both “must-have” and “risk-control” items. Prices are approximate and time-sensitive; supplier links are via the cited official pages where possible.
| BOM Line Item | Example Product / Spec | Supplier (Priority: Official) | Qty | Est. Cost (USD) |
|---|---|---|---|---|
| LED grow light (2x4 class) | IONBOARD S24, 220 W | AC Infinity [30] | 1 | $239 |
| LED grow light (2x2 class, alternative) | IONBOARD S22, 115 W | AC Infinity [30] | 1 | $119 |
| Inline ventilation kit (optional but strong for apartments) | Air Filtration Kit 4" | AC Infinity [30] | 1 | $159 |
| Grow tent (optional containment) | CLOUDLAB 422 (2x2) or larger | AC Infinity [30] | 0–1 | $129–$239 (size-dependent) |
| High reliability pH/EC meter | Combo Meter | Bluelab [35] | 1 | $356 |
| High-pressure diaphragm pump (HPA path) | 8800 Series diaphragm pump (spec sheet available) | Aquatec [36] | 1 | (varies) |
| Pump datasheet anchor | 8800 Series 24VAC ~2.6 LPM spec | Aquatec [36] | — | — |
| Misting assemblies (low-flow, check valve) | Value L Misting Assembly | MistKing [37] | 3–6 | (varies) |
| Agricultural spray nozzle reference (alt) | Catalog (ConeJet / TX series reference) | TeeJet Technologies [38] | — | — |
| Ultrasonic fogger (fogponics) | 5-disc mist maker ~1900 ml/hr | The House of Hydro [39] | 0–1 | (varies) |
Supplier/source anchors: AC Infinity official product listings for IONBOARD series lights and tents. [40] Bluelab official price listing for the Combo Meter. [41] Aquatec official pump page and a published spec sheet for the 8800 pump series. [42] MistKing official nozzle assembly page. [43] TeeJet catalog PDF as a primary spec reference for spray components. [44] House of Hydro product page as a fogger example. [45]
Environmental impacts: life-cycle framing and DIY implications
Peer-reviewed comparative findings (hydroponic CEA vs field supply chains). An attributional LCA for lettuce supply chains found hydroponic CEA used ~15 kWh electricity per kg lettuce supplied, and under fossil-heavy grids this can yield high footprints (example up to ~17.8 kg CO₂e/kg lettuce in their scenario set), while renewable electricity (and land-use opportunity cost assumptions) can reduce footprints substantially (example ~0.48 kg CO₂e/kg lettuce in one renewable case). [2]
Modeled field vs greenhouse hydroponic resource tradeoff (water vs energy). A quantitative comparison for lettuce found hydroponics had much higher yield per area and far lower direct water per kg than conventional production but required far more energy per kg (reported energy ratio ~82×). [3]
Implication for DIY aeroponics. Aeroponics may reduce direct water losses versus some hydroponic configurations (because no standing root bath), but in full‑indoor personal systems, lighting energy dominates and can overwhelm water gains from a life-cycle perspective unless electricity is low‑carbon. This follows directly from the LCA hotspot results emphasizing electricity for lighting/climate control. [32]
Operations, Maintenance, Food Safety, and Compliance
Maintenance schedules and labor estimates (practical baseline)
Aeroponics functions like a small process plant: short-cycle pumping + nutrient chemistry + sanitation. The most common DIY failure modes map directly to what the literature highlights: droplet deposition uniformity, nozzle performance, nutrient solution chemistry changes during atomization, and microbial/biofilm dynamics in recycled solutions. [46]
A realistic maintenance cadence for a 4–12 plant leafy greens module (time estimates are planning values, not universal):
· Daily (3–8 minutes): verify pump operation (audible/visual), check reservoir level, quick plant inspection.
· 2–4× per week (10–20 minutes): measure and adjust pH/EC; top up water; wipe splash/condensation surfaces. pH/EC monitoring is central to nutrient uptake control; guidance emphasizes frequent measurement and adjustment. [47]
· Weekly (20–45 minutes): clean prefilters/strainers; inspect nozzles for clogging; verify timer settings and backup behavior. The engineering literature highlights that nozzle parameter uncertainty and clogging/overlap can confound performance and reproducibility. [7]
· Every crop cycle or monthly (1–3 hours): drain and clean reservoir/root chamber surfaces, sanitize or swap lines/nozzles as needed, recalibrate instruments.
Food safety and pest/pathogen risks
Even at home scale, the relevant hazard classes are: human / plant pathogens, and chemical/physical contamination.
A major extension resource on hydroponic/aquaponic produce safety highlights why soilless systems are unique:
· The nutrient solution is nutrient-rich, which can support pathogen survival/growth if contaminated. [48]
· Many systems have circulation, meaning contamination can spread rapidly throughout the system. [48]
· Non-soil surfaces (rafts, troughs, structural surfaces) become de facto food contact surfaces due to plant contact and handling; cleaning and sanitation procedures and hygienic design are emphasized. [48]
While those resources focus on hydroponics broadly, the same logic applies to aeroponic reservoirs and return plumbing: the water connects the system.
Plant disease dynamics can also be rapid in recirculating systems. An extension hydroponics fact sheet notes that diseases such as fusarium and verticillium can spread quickly through the system, underscoring the need for early detection and sanitation. [6]
Risk mitigations (high value controls):
· Use a clean water source (often reverse osmosis) and maintain pH/EC in recommended ranges; engineered aeroponic systems may dose from RO water and concentrated nutrient stock. [17]
· Treat reservoir and any surface contacted by recirculated solution as a cleanable food-contact surface; design for tool access and cleaning. [48]
· Prioritize filtration and clog control; atomization is a critical control point, and droplet coalescence/clogging changes droplet size distributions and deposition. [9]
· Manage humidity to reduce condensation drip pathways and microclimate pathogen pressure; vertical/shelf systems require explicit heat/humidity planning. [16]
Risk mitigations (high value controls):
· Use a clean water source (often RO in engineered systems) and maintain pH/EC in recommended ranges; engineered aeroponic systems may dose from RO water and concentrated nutrient stock. [17]
· Treat reservoir and any surface contacted by recirculated solution as a cleanable food-contact surface; design for tool access and cleaning. [48]
· Prioritize filtration and clog control; atomization is a critical control point, and droplet coalescence/clogging changes droplet size distributions and deposition. [9]
· Manage humidity to reduce condensation drip pathways and microclimate pathogen pressure; vertical/shelf systems require explicit heat/humidity planning. [16]
Scalability, Modular Upgrades, Timeline, and Recommendations
Scalability and modular upgrade paths
A staged approach reduces risk by validating each control layer before adding complexity—aligned with the literature’s emphasis on confounding variables in aeroponic system configuration and the need for reproducible design choices. [9]
Recommended modular path
· Module 1 (4 plants): single chamber + stable lighting + basic pH/EC measurement + conservative spray schedule. Demonstrate ≥2 consecutive successful crop cycles.
· Module 2 (12 plants): scale canopy area (often 2×4) and add ventilation containment; introduce redundancy (dual timers, reservoir level alarm).
· Module 3 (multi-module): duplicate chambers rather than growing one large chamber; this limits “blast radius” from contamination or pump failure and enables A/B testing of spray cycles and nutrient regimes.
High-leverage upgrades
· Atomization reliability upgrades: filtration, check valves, better manifold design, and pressure stabilization; engineering designs explicitly pair pressure swirl atomizers with pressured tanks and solenoid valve control of spray cycles. [49]
· Measurement upgrades: move from hobby pens to reliable pH/EC instruments; a professional-grade meter is a major risk reducer for nutrient drift. [50]
· Lighting optimization upgrades: choose fixtures with clear PPFD maps and µmol/J ratings; extension guidance recommends avoiding lights without spectrum graph/PPFD information and explicitly uses µmol/J for efficiency comparisons. [27]
· Sanitation upgrades: design-for-cleaning (smooth surfaces, tool access) and documented SOPs; the produce-safety guidance emphasizes SOPs and hygienic design principles because many systems are not designed for cleaning. [48]
12-month setup and optimization timeline
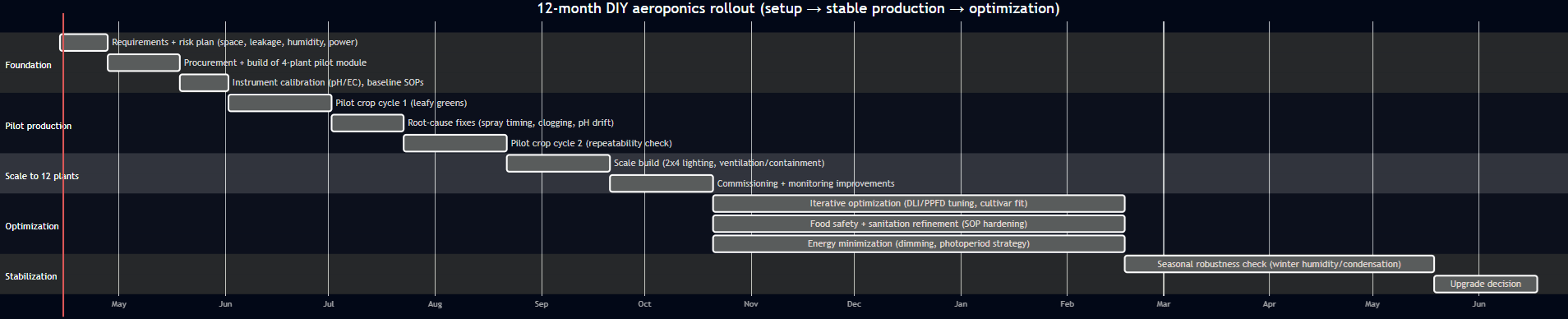
This roadmap reflects two evidence-backed realities: (i) yields depend strongly on correct droplet delivery/timing, implying iterative commissioning, and (ii) electricity dominates environmental performance in full-indoor systems, implying sustained optimization of lighting and climate. [51]
Recommendations
1) Default to leafy greens and herbs for the first 3–6 months, because they match lower DLI/PPFD requirements and shorter cycles, enabling faster learning loops. Lettuce DLI and PPFD guidance supports this lower-energy starting set relative to fruiting crops. [52]
2) Design for sanitation access and treat recirculating-solution surfaces as food-contact surfaces, even at home scale. This single decision reduces both human pathogen and plant pathogen risk and aligns with extension produce-safety guidance for soilless operations. [53]
3) Invest early in measurement and reliability rather than “more plants.” A high-quality pH/EC meter and stable lighting are risk-reducing CAPEX items; yield losses from pH mismanagement and misting misconfiguration are documented concerns. [54]
4) Treat electricity as the binding constraint and optimize for µmol/J and DLI delivery efficiency. The LCA literature and extension lighting guidance converge on the conclusion that lighting/climate power dominates footprints and costs; therefore, dimming control, good fixture selection, and avoiding excess PPFD are the highest-ROI levers. [55]
Key References
· Aeroponic system definition and timer cadence (extension). [6]
· Engineering design considerations: droplet size targets, atomizer types, pressure tank + solenoid control, nozzle selection realities. [56]
· Aeroponic yield sensitivity to droplet size and spray interval (butterhead lettuce). [57]
· Aeroponic physiology and system model (deposition/retention/decay), productivity examples for multiple crops, and identified knowledge gaps (peer‑reviewed review). [58]
· Produce safety considerations for soilless systems (water as a contamination vector; circulation; food-contact surfaces; hygiene/SOPs). [48]
· Lighting targets for lettuce: DLI and PPFD guidance; LED efficiency framing. [59]
· Environmental footprint benchmarking for lettuce (hydroponic CEA vs field supply chains; energy per kg; dependence on electricity mix). [60]
[1] [6] Fact Sheet - Hydroponics
https://extension.okstate.edu/fact-sheets/hydroponics
[2] [28] [32] [55] [60] Comparative environmental footprints of lettuce supplied by hydroponic controlled-environment agriculture and field-based supply chains
[3] [33] Comparison of Land, Water, and Energy Requirements of Lettuce Grown Using Hydroponic vs. Conventional Agricultural Methods
[4] [7] [9] [10] [11] [13] [17] [18] [35] [36] [38] [39] [49] [56] Aeroponic systems design: considerations and challenges
https://www.agroengineering.org/jae/article/download/1387/1063/9776
[5] [16] [21] [27] [37] Controlled Environment Agriculture: Understanding Grow Lights
https://extension.missouri.edu/publications/g6987
[8] [12] [23] [46] [58] Getting to the roots of aeroponic indoor farming
https://data.nbi.ac.uk/systems/OpenAccess/65288
[14] [15] [25] [26] [51] [57] Effects of droplet size and spray interval on root-to-shoot ratio, photosynthesis efficiency, and nutritional quality of aeroponically grown butterhead lettuce
https://pdfs.semanticscholar.org/8acc/d7382ec0abda95d5166da0271e16890bb3a4.pdf
[19] [29] [30] [52] [59] Calculating and Using Daily Light Integral (DLI): An Introductory Guide
https://www.pubs.ext.vt.edu/SPES/spes-720/spes-720.html
[20] [47] [54] Test & Adjust Nutrients in Hydroponics
https://e-gro.org/pdf/E503.pdf
[22] Yield and nutritional quality of aeroponically cultivated basil as affected by the available root-zone volume
https://ejfa.me/index.php/journal/article/download/984/715/1456
[24] Determining the Economic Value of Providing Supplemental Light to Lettuce During Winter Production
https://www.extension.purdue.edu/extmedia/HO/HO-283-W.pdf
[31] [40] AC Infinity Grow Lights
https://acinfinity.com/grow-lights/
[34] [48] [53] Produce Safety in Hydroponic and Aquaponic Operations
https://www.uvm.edu/extension/necafs/produce-safety-hydroponic-and-aquaponic-operations
https://bluelab.com/products/bluelab-combo-meter
[42] Aquatec Pressure Booster Pumps
https://www.aquatec.com/pumps/pressureboosterpumps.html
[43] Mistking Value L Misting Assembly
https://mistking.eu/Value-L-Misting-Assembly.html
[44] TeeJet Technologies Catalog 51A-M
https://www.teejet.com/en/-/media/dam/agricultural/usa/sales-material/catalog/cat51a_metric.pdf
[45] The House of Hydro - 5 Disc Mist Maker with Float and Spare Discs
https://thehouseofhydro.com/products/5-disc-mist-maker-with-float-and-spare-discs
Our site has zero commercial incentive or affiliation with any retailers listed above.





