Perfluorocarbon Liquid Breathing: High-G Tolerance, Saturation Diving Beyond 700 msw, and the DARPA-to-FDA Path
PFC liquid ventilation, HPNS, G-LOC, Inolivent, Orixha, NEDU, and 711th HPW: what the science supports and what remains unvalidated in humans.

Perfluorocarbon Liquid Breathing: The Path to High-G Tolerance and Deep Saturation Diving
1. Summary
Perfluorocarbon (PFC) liquid breathing has occupied a peculiar position in the human performance literature for nearly six decades: scientifically plausible, repeatedly demonstrated in mammalian models, and yet to date never validated in controlled adult human trials for the two domains where it would have the most transformative consequences, namely sustained high-acceleration aviation and extreme-depth saturation diving. The foundational experiments of Clark and Gollan in 1966, in which mice and cats survived prolonged immersion in oxygenated fluorocarbon liquids, established that mammalian gas exchange across the alveolar-capillary membrane could occur in the absence of a gas phase [1]. Subsequent work by Kylstra at Duke University, by the Shaffer and Wolfson research group at Temple University, by Hirschl and colleagues at the University of Michigan, and most recently by the Inolivent consortium at the Université de Sherbrooke has refined the engineering of partial liquid ventilation (PLV) and total liquid ventilation (TLV) to a point where clinical-grade prototypes exist for animal subjects up to roughly 80 kilograms in body mass [2][3][11].
For the high-G domain, the theoretical case is that filling the lung with an incompressible perfluorocarbon would eliminate the air-liquid interface that is the proximate cause of acceleration-induced atelectasis and would, in combination with whole-body fluid immersion, permit hydrostatic pressure equalization across the thorax at acceleration loads well beyond the present operational ceiling of approximately +9 Gz sustained by pilots wearing anti-G suits and performing the anti-G straining maneuver [19][20]. The available human data on this application are essentially absent. The European Space Agency's Advanced Concepts Team has explored the concept analytically, but no peer-reviewed centrifuge experiment in humans using PFC ventilation has been published, and the present report does not identify any open-source military centrifuge program that has progressed to manned trials.
For the deep-diving domain, helium-oxygen and hydrogen-helium-oxygen mixtures have allowed simulated chamber dives to a maximum of 701 metres of seawater (msw), reached by COMEX diver Théo Mavrostomos in the Hydra X experiment of 1992, with offshore working depth records around 534 msw [16]. Beyond this range, high-pressure nervous syndrome (HPNS), gas density, and respiratory work limit further descent. PFC-filled lungs would in principle eliminate gas-phase compression and inert gas narcosis effects, but the experimental record in mammals indicates that carbon dioxide elimination, not oxygenation, is the rate limiting constraint, and no human exposure under hyperbaric conditions with PFC ventilation has been reported in the open literature [2][14].
The institutional landscape is bifurcated. Defense-aerospace research is concentrated in the U.S. Air Force Research Laboratory's 711th Human Performance Wing, the U.S. Navy Experimental Diving Unit, the DARPA Biological Technologies Office, and allied research bodies operating under NATO's Science and Technology Organization and the AUKUS Pillar 2 undersea capabilities working group [22][26][28]. The clinical and engineering base resides in academic centers and a small number of start-ups, notably Orixha (a Sherbrooke and Créteil spinout founded by the Tissier-Micheau group) and OriGen Biomedical, which acquired the LiquiVent dataset from Alliance Pharmaceutical in 2013 after Alliance discontinued the program [13]. The primary technical barriers remain CO₂ washout under TLV, ventilator miniaturization, transition-back-to-gas pulmonary clearance, and the absence of a regulatory pathway for non therapeutic enhancement use. Near-term investment considerations are driven less by clinical respiratory indications than by adjacent applications such as ultra-rapid therapeutic hypothermia, where TLV has shown reproducible neuroprotective effects in cardiac-arrest models [13][21]. Policymakers should expect a 7-to-15-year horizon before any operational defense application becomes plausible, with the regulatory gating step likely arising under investigational frameworks rather than full commercial drug approval.
- Summary
- Contextual Background and Scientific Foundations
- 2.1 Biophysics of Liquid Breathing
- 2.2 Historical Development and Research Milestones
- 2.3 PFC Chemistry and Candidate Compounds
- Key Stakeholders and Institutional Landscape
- 3.1 Defense and Aerospace Medical Research Programs
- 3.2 Academic and Clinical Research Centers
- 3.3 Commercial and Industrial Actors
- 3.4 Diving and Undersea Medicine Community
- Technical and Operational Considerations
- 4.1 High-G Tolerance: Physiological Problem
- 4.2 Liquid Breathing as High-G Countermeasure: Theoretical Mechanisms
- 4.3 Engineering Challenges: The Liquid Ventilator
- 4.4 Deep Saturation Diving: Physiological Problem
- 4.5 Liquid Breathing for Extreme-Depth Diving: Mechanisms and Evidence
- 4.6 Human Factors and Interface Challenges
- Economic and Market Dynamics
- 5.1 Defense Procurement and Investment Signals
- 5.2 Commercial Saturation Diving Market
- 5.3 Pharmaceutical and Medical Device Pathways
- 5.4 Dual-Use Technology Valuation
- Regulatory Landscape
- 6.1 Medical Device and Drug Approval Pathways
- 6.2 Military Exemptions and Investigational Use Frameworks
- 6.3 Occupational Safety and Diving Regulations
- 6.4 Ethical and Consent Considerations
- Geopolitical and Strategic Dimensions
- 7.1 Strategic Value of Extended Human Performance Envelopes
- 7.2 Comparative National Programs
- 7.3 Alliance and Technology Sharing Implications
- 7.4 Undersea Strategic Competition
- Risk Matrix
- Strategic Recommendations
- 9.1 Audience A: Defense Policymakers and Military Program Offices
- 9.2 Audience B: Institutional Investors and Deep-Sea Industry Executives
- References
2. Contextual Background and Scientific Foundations
2.1 Biophysics of Liquid Breathing
The physical premise of liquid ventilation rests on the very high gas solubility of fluorinated organic liquids. PFCs dissolve oxygen in quantities roughly twenty times greater than water and carbon dioxide approximately three times greater, with dissolution following Henry's law in a strictly linear, non-cooperative manner [9]. In a liquid-ventilated lung, oxygen and carbon dioxide exchange occurs by simple diffusion between the perfluorocarbon film coating the alveolar surface and the pulmonary capillary blood, with no air-liquid interface and therefore no surface tension at the alveolar wall [3][5]. The available data indicate that adequate arterial oxygenation is readily achieved under both partial liquid ventilation, in which the lung is filled to functional residual capacity (FRC) with PFC and gas-tidal ventilation continues, and total liquid ventilation, in which liquid tidal volumes are mechanically delivered and withdrawn by a dedicated liquid ventilator [3][11].
The principal physiological challenge is carbon dioxide elimination. Because PFC viscosity is one to two orders of magnitude greater than that of air and PFC density approaches twice that of water, the work of breathing rises sharply, and tidal volumes must be cycled at flows on the order of several litres per minute to match resting metabolic demand [1][2]. Clark and Gollan reported partial CO₂ retention in cats even at optimal conditions, and Kylstra's 1962 experiments with hyperbaric oxygenated saline produced respiratory acidosis sufficient to kill the animals despite adequate oxygenation [1][2]. Modern engineering approaches, particularly those of the Inolivent group, have addressed CO₂ washout by precise control of expiratory flow profiles and by independent piston pumps that prevent expiratory airway collapse [11]. The current literature does not establish a CO₂ washout solution that is robust at the metabolic rates of an active adult human under sustained physical or physiological stress.
2.2 Historical Development and Research Milestones
The trajectory of liquid ventilation research moves through three identifiable phases. The first, from the early 1960s through the late 1970s, was dominated by the demonstration experiments of Kylstra at Duke and Leiden, of Clark and Gollan at the University of Cincinnati, and of Modell, Newby, and Ruiz on long-term survival following fluorocarbon ventilation [1]. The U.S. Office of Naval Research funded much of this work in the context of submarine escape and extreme depth diving, and the period included the documented exposure of commercial diver Frank Falejczyk to oxygenated saline in a clinical setting under Kylstra's supervision; Falejczyk subsequently developed pneumonia attributable to incomplete liquid extraction from his lungs.
The second phase, from approximately 1989 through the early 2000s, was driven by neonatal and adult respiratory medicine. Greenspan, Wolfson, and Shaffer published the first liquid ventilation of human preterm neonates in 1989-1990 [31]. Leach and colleagues' 1996 New England Journal of Medicine report described partial liquid ventilation with perflubron in 13 premature infants with severe respiratory distress syndrome refractory to surfactant, conducted as a phase I-II study under an Alliance Pharmaceutical-sponsored Investigational New Drug (IND) application; eight of thirteen infants survived to 36 weeks corrected gestational age, and arterial oxygen tension increased 138 percent within one hour of perflubron instillation [4]. Hirschl and colleagues extended the technique to adults, first in a multicenter phase I-II non controlled trial reported in 1998 and subsequently in a randomized controlled pilot study published in 2002 in which 90 adults with acute respiratory distress syndrome (ARDS) received either PLV with perflubron or conventional mechanical ventilation [6][7]. Although early observational data suggested improvements in compliance and oxygenation, the controlled study did not demonstrate a mortality benefit superior to conventional protective ventilation, and a subsequent multicenter phase III trial (Kacmarek and colleagues, published 2006) was terminated for similar reasons [8]. Alliance Pharmaceutical discontinued the LiquiVent program for ARDS indications in May 2001, and the trial dataset was acquired by OriGen Biomedical in 2013.
The third and current phase, from roughly 2005 to the present, is characterized by the Inolivent consortium's iterative development of TLV prototypes (Inolivent-3 through Inolivent-9) and by the application of TLV to ultra-rapid therapeutic hypothermia for cardiac arrest, primarily by the Tissier-Berdeaux-Ghaleh group at INSERM U955 in collaboration with Sherbrooke [11][13][21] [29]. The clinical failure of perflubron in ARDS has had two implications for the high-G and deep-diving applications: first, it removed the most plausible commercial path that would have generated regulatory approval, manufacturing scale, and operator training; second, it preserved a substantial body of safety data on perflubron exposure that may shorten any future investigational pathway.
2.3 PFC Chemistry and Candidate Compounds
Perfluorocarbons are organofluorine compounds in which all carbon-bound hydrogen atoms have been replaced with fluorine. The carbon-fluorine bond's strength and the weak intermolecular forces between fluorinated chains give the resulting liquids exceptional chemical inertness, low surface tension (15 to 20 dynes per centimeter at 25°C), high density (1.75 to 1.95 grams per millilitre), low water solubility, and high respiratory gas solubility [5][9]. The two compounds most extensively studied for pulmonary use are perflubron (perfluorooctyl bromide, C₈F₁₇Br, molecular weight 499) and perfluorodecalin (C₁₀F₁₈). Perflubron has higher vapor pressure (approximately 11 torr at 37°C), which permits more rapid pulmonary clearance by evaporation but also requires higher dosing frequency during PLV; perfluorodecalin has lower vapor pressure but distributes less homogeneously and has been associated with hyperinflated lung syndrome in some animal models [5][10].
Available data indicate that perflubron is biocompatible, is not metabolized, and is eliminated principally by exhalation as vapor and by transcutaneous loss, with residence times in the lung extending from days to years depending on dose and ventilation strategy. A 2018 case report documented "pseudo-calcifications" on a computed tomography scan 15 years after perflubron exposure, indicating that small residues can persist for the entire post-exposure life of clinically relevant patients without apparent clinical consequence; this finding is reassuring for safety but presents specific concerns for radiological signature and for personnel who may need to operate in mixed-environment scenarios. Tissue accumulation in the reticuloendothelial system following intravenous emulsion exposure is documented in the perfluorocarbon blood substitute literature; the relevance to inhaled neat liquid is debated and the current literature does not establish a quantitative chronic-toxicity threshold for adult human pulmonary exposure [9][10].
3. Key Stakeholders and Institutional Landscape
3.1 Defense and Aerospace Medical Research Programs
The U.S. Department of Defense remains the largest organizationally coherent stakeholder in extreme human performance research relevant to liquid breathing. Within the U.S. Air Force, the 711th Human Performance Wing at Wright-Patterson Air Force Base operates the only currently fielded human-rated centrifuge in the U.S. Air Force and manages aerospace medicine research through the U.S. Air Force School of Aerospace Medicine (USAFSAM); the wing's research portfolio includes acceleration physiology, G-LOC mitigation, and centrifuge protocols for fighter and astronaut training [referenced in 711 HPW open documentation]. The wing's 9G-capable centrifuge and its biodynamics laboratory (which includes a horizontal impulse accelerator and vertical deceleration tower) provide the physical infrastructure that any liquid-breathing acceleration trial would require.
The U.S. Navy Experimental Diving Unit (NEDU) at Panama City, Florida, is the primary federal diving and hyperbaric research command. The NEDU houses the Ocean Simulation Facility, the largest manned hyperbaric research complex in the world, and conducts at least one saturation dive per year for evaluation of decompression schedules, breathing gases, life-support equipment, and thermal protection. The NEDU absorbed the diving biomedical and development division of the former Naval Medical Research Institute in 1998. The Naval Medical Research Unit Dayton (NAMRU-Dayton, formerly Naval Aerospace Medical Research Laboratory) co-located with the 711th HPW, provides naval aerospace medical research continuity. DARPA's Biological Technologies Office, established in 2014, has issued broad agency announcements that include human performance optimization and biomedical research areas relevant to liquid breathing, although no public-record DARPA program names liquid ventilation as a specific objective [22]. NASA's Human Research Program addresses sustained acceleration in the context of launch and reentry, particularly for commercial-crew vehicles with steeper return trajectories; NASA personnel have used the 711th HPW centrifuge for astronaut acceleration training under interagency agreements.

3.2 Academic and Clinical Research Centers
The principal academic loci of liquid ventilation research are Temple University School of Medicine in Philadelphia, where Marla R. Wolfson and Thomas H. Shaffer have produced the longest continuous program of physiological and translational PFC research; the Université de Sherbrooke in Quebec, where the Inolivent group (Hervé Walti, Philippe Micheau, Raymond Robert, and clinical-pediatric collaborators including Jean-Paul Praud) has developed the Inolivent-3 through Inolivent-9 ventilator prototypes [11]; the Université Paris-Est Créteil and the École Nationale Vétérinaire d'Alfort, where Renaud Tissier, Matthias Kohlhauer, Bijan Ghaleh, and Alain Berdeaux have pursued TLV-mediated therapeutic hypothermia [13][21][29]; the University of Michigan, associated with Hirschl's adult ARDS work [6][7]; and the State University of New York at Buffalo's Center for Research and Education in Special Environments (CRESE), historically associated with Lundgren and Fuhrman. The Sherbrooke and Créteil groups jointly hold the principal patent portfolio in modern total liquid ventilation, including U.S. Patent 7,726,311 and U.S. Patent 9,439,804.
3.3 Commercial and Industrial Actors
The most consequential commercial actor in the history of PFC respiratory medicine was Alliance Pharmaceutical Corporation of San Diego, the originator and developer of LiquiVent (perflubron) and of the related intravenous oxygen carrier emulsion Oxygent. Alliance entered a 1996 license agreement with Hoechst Marion Roussel for LiquiVent that was terminated in 1998 when development complexity exceeded projections, and Alliance ultimately discontinued LiquiVent for acute lung injury, ARDS, and neonatal respiratory distress syndrome on 29 May 2001 following phase III results that did not show superiority over high-frequency oscillatory ventilation. OriGen Biomedical of Austin, Texas, acquired the LiquiVent clinical trial dataset and trademark in June 2013, stating an intent to support future regulatory submissions; no publicly reported FDA filing has resulted to date. Orixha, a French startup co-founded by Tissier, Kohlhauer, Micheau, Berdeaux, Walti, and Nadeau, is the principal current commercial entity pursuing TLV, with an indication focus on cardiac arrest and post-resuscitation neuroprotection rather than respiratory failure [13]. No publicly verifiable commercial entity is at present pursuing PFC ventilation for high-G tolerance or extreme-depth diving applications, and any claim to the contrary should be regarded as unsubstantiated absent disclosed regulatory filings or peer-reviewed reports.
3.4 Diving and Undersea Medicine Community
The commercial saturation diving community is organized under the International Marine Contractors Association (IMCA), whose code of practice (IMCA D 014) and Diving Equipment Systems Inspection Guidance (IMCA D 024 for saturation systems) constitute the de facto international operational standard [24]. Medical guidance is issued by the Diving Medical Advisory Committee (DMAC), supported by IMCA, with DMAC 15 (medical equipment and drug list) and DMAC 28 (saturation diving medical support) as the principal references [25]. Operationally, the saturation diving fleet is concentrated among Subsea 7, TechnipFMC, DOF Subsea, Helix Energy Solutions (including the Seawell, with a saturation system rated to 300 metres), Oceaneering International, Aqueos Corporation (which introduced an AMSS Modular Saturation Diving System in recent years), and a number of regional operators including K Subsea, NDE Offshore, and Mermaid Subsea Services. The historical scientific lead in extreme depth saturation research was held by COMEX of Marseille, which conducted the Hydra series of experiments from 1968 to 1996, including Hydra V (450 msw, 1985), Hydra VIII (the 534 msw open-sea pipeline-connection record of 1988), and Hydra X (the 701 msw chamber dive of November 1992) [16]. The principal investigators in HPNS research, Peter Bennett (Duke University) and Jean-Claude Rostain (CNRS Marseille), produced the foundational characterization of the syndrome that constitutes the present physiological barrier to deeper saturation diving [14][17].

4. Technical and Operational Considerations
4.1 High-G Tolerance: Physiological Problem
Sustained positive z-axis acceleration (+Gz) induces a head-to-toe gravito-inertial gradient that displaces blood toward the lower body, reduces cerebral perfusion pressure, and produces a sequence of greyout, blackout, and finally G-induced loss of consciousness (G-LOC). Without protection, unaugmented +Gz tolerance is approximately +4 to +5 Gz; with a properly fitted anti G suit, anti-G straining manoeuvre, and pressure breathing for G (PBG), trained fighter pilots can sustain peak +Gz of approximately +9 Gz for short intervals [19][20]. Sevilla and colleagues reported that 72 percent of 74 G-LOC mishaps in their dataset were directly related to inadequate AGSM performance; a study at the Indian Air Force Institute of Aerospace Medicine using a high-performance human centrifuge documented G-LOC and almost-loss-of-consciousness events even among trained aircrew at sustained 9G profiles [referenced in centrifuge literature]. The physiological ceiling is determined principally by cardiovascular factors, although alveolar collapse and ventilation-perfusion mismatch under sustained acceleration also contribute, particularly above +6 Gz where pulmonary atelectasis becomes radiographically demonstrable.
4.2 Liquid Breathing as High-G Countermeasure: Theoretical Mechanisms
The conceptual basis for PFC liquid breathing as a high-G countermeasure rests on two interlocking arguments. First, an incompressible, near-isodense medium filling the lungs would eliminate the gas-phase compression and atelectasis that contribute to ventilation-perfusion mismatch under acceleration. Second, full immersion of the body in a similar-density fluid, combined with PFC-filled lungs, would in principle equalize hydrostatic pressure across the thorax and abdomen, removing the cardiovascular gradient that drives G-LOC. The European Space Agency's Advanced Concepts Team has analytically explored a fluid-immersion concept that posits sustainable acceleration tolerance well above 24G if both lung filling and external immersion are achieved.
The available animal experimental evidence for this hypothesis is limited and largely indirect. Studies of liquid-filled lungs in centrifuge-exposed small mammals have been conducted, but no peer-reviewed report identifies a controlled large-animal experiment using TLV with simultaneous high-G centrifuge exposure and acceleration tolerance as a primary endpoint. The published lung-physiology literature on +Gz exposure in humans (for example the King's College London centrifuge protocol on advanced gravitational lung physiology) examines gas ventilated subjects only. This remains an area of active speculation rather than established physiology, and any operational claim that liquid breathing has been "demonstrated" to extend human acceleration tolerance is unsupported by peer-reviewed evidence as of this report.
4.3 Engineering Challenges: The Liquid Ventilator
A clinical-grade total liquid ventilator must oxygenate and decarbonate a continuously circulated PFC stream, deliver and withdraw tidal liquid volumes at programmed rates and pressures, condition the temperature of the inspirate, recover evaporative PFC losses, and prevent expiratory airway collapse. The Inolivent-4 prototype incorporates two independent piston pumps, a membrane oxygenator, an integrated heating system, a buffer reservoir, and a condenser to recapture vapor losses; it has been used in animal experiments on subjects from 460 grams to 79.5 kilograms in body mass, including newborn lambs and large pigs [11]. The Inolivent-9 represents the most recent prototype optimized for neonatal intensive care applications. The Sherbrooke group, Tissier and colleagues at Créteil, and Orixha have demonstrated successful TLV transitions in 13-year-old non-human primates and in adult-sized pigs with subsequent return to spontaneous gas breathing and ten-day follow-up showing no macroscopic perfluorocarbon retention on thoracic computed tomography [13]. The gap between this laboratory capability and operational deployment in a field environment is substantial. A deployable system would require ruggedization, reduction in mass and volume to perhaps one-quarter of the current bench prototypes, integration with portable PFC supply, and extensive fail-safe engineering for the catastrophic case of pump failure or oxygenator embolism.
4.4 Deep Saturation Diving: Physiological Problem
Conventional helium-oxygen (heliox) saturation diving below approximately 150 to 200 metres of seawater encounters HPNS, a syndrome of tremor, vertigo, nausea, EEG slow-wave activity, sleep disruption, and cognitive impairment that was first described by Brauer and colleagues in 1969 [15] and characterized in subsequent work by Bennett and Rostain [14][17]. HPNS is caused by the hydrostatic pressure itself rather than by helium per se, and its severity depends on rate of compression, depth attained, and gas mixture. Two principal mitigation strategies are slowed compression and the addition of a narcotic gas (small percentages of nitrogen in trimix, or hydrogen in hydreliox) to partially reverse the pressure-induced excitation. The COMEX Hydra series demonstrated that hydreliox extended workable depth into the 500 to 600 msw range, with the 1988 Hydra VIII offshore dive reaching 534 msw and the 1992 Hydra X chamber dive reaching 701 msw, the latter under conditions in which two of three divers were "incapacitated" at 675 msw and only Mavrostomos completed a brief two-hour excursion to 701 msw [16]. The fundamental physical limits below this depth are the work of breathing imposed by gas density and the unmitigated HPNS phenomenon.
4.5 Liquid Breathing for Extreme-Depth Diving: Mechanisms and Evidence
PFC liquid breathing is theoretically attractive for extreme-depth applications because the incompressible PFC has no gas-phase compression behavior, eliminates inert gas narcosis, and removes the gas-density work-of-breathing constraint. The historical experimental record consists principally of Kylstra's 1962 and subsequent work demonstrating mammalian survival under hyperbaric oxygenated saline, and Lundgren's research on respiratory mechanics under hyperbaric conditions at SUNY Buffalo. Lynch and colleagues conducted hyperbaric liquid breathing experiments in dogs in the early 1980s. The dominant finding across all this work is that CO₂ retention rather than O₂ delivery is the limiting factor, because the kinematic viscosity of PFC is approximately 5 to 10 times that of water and CO₂ diffusivity in PFC, while higher than in saline, does not approach the values needed to clear the metabolic CO₂ load of an actively working diver under realistic conditions [1][2].
A separate question concerns whether liquid breathing would address HPNS, given that the pressure-induced neurological excitation is mediated principally at the cellular and synaptic level (with NMDA receptor and voltage-gated calcium channel modulation as principal mechanisms) rather than by gas-phase phenomena. The current literature does not establish whether eliminating gas exchange would alter HPNS susceptibility in a measurable way. Distinguishing theoretical projections from experimentally verified results, the evidence supports the claim that PFC ventilation in principle removes gas-phase compression effects but does not support any claim that humans have successfully descended beyond conventional saturation limits using PFC ventilation.
4.6 Human Factors and Interface Challenges
The psychological dimensions of voluntary liquid inhalation have not been systematically studied in adult human subjects. The historical demonstration of liquid breathing in commercial diver Frank Falejczyk under Kylstra's supervision produced pneumonia and significant operational morbidity. The film "The Abyss" (1989) brought public attention to the concept; its director consulted with Kylstra and Bennett but used animal subjects (rats) for the demonstration scene. No controlled adult human TLV trial has been conducted for high-G or deep-diving applications. Training protocols, transition-induction procedures, and emergency reversion to gas breathing all remain undefined for these use cases. Voice communication, manual dexterity in liquid-filled airways, and post-exposure neurocognitive recovery represent operational unknowns that would require substantial dedicated study before any field deployment could be considered.

5. Economic and Market Dynamics
5.1 Defense Procurement and Investment Signals
Aggregate defense procurement signals in human performance and life-support technology constitute the most relevant baseline against which any liquid-breathing investment case should be measured. Anti-G suit modernization, advanced ejection seats (notably the Martin-Baker US16E in F-35 and successor seats), pilot physiological monitoring systems, and aerospace medicine research collectively represent a procurement and research, development, test and evaluation (RDT&E) line of several hundred million U.S. dollars annually across NATO air forces. DARPA's Biological Technologies Office RDT&E budget supports broad agency announcements that have historically included human performance optimization research areas, although no public solicitation specifically names liquid breathing or perfluorocarbon ventilation as a program element [22]. The 711th HPW centrifuge investment and the NEDU Ocean Simulation Facility represent the test-and-evaluation infrastructure that any liquid-breathing program would draw upon.
5.2 Commercial Saturation Diving Market
The commercial saturation diving services market is small relative to the broader offshore energy economy but strategically concentrated. Industry market-research estimates place the diving support vessel market at approximately USD 4.2 billion by 2033, with the saturation diving services market segment growing at single-digit percentages and concentrated among the operators identified in section 3.4. Saturation diving accounts for approximately 21 to 24 percent of commercial diving service-type revenue in published industry analyses, with most assignments occurring in the 100 to 300 msw range. The economic value of extending depth limits depends critically on whether subsea infrastructure (oil and gas, telecommunications cables, offshore wind cabling, deep-sea mining) at depths beyond present saturation limits requires manned intervention; current trends favor remotely operated vehicles and autonomous systems for tasks below approximately 300 msw, suggesting that the commercial pull on a liquid-breathing technology would be modest unless paired with a defense-driven base-load demand. These market estimates derive from commercial market-research publications and should be treated as indicative rather than authoritative.
5.3 Pharmaceutical and Medical Device Pathways
The cost and timeline of bringing a new PFC respiratory compound through a U.S. Food and Drug Administration (FDA) approval pathway can be approximated from the LiquiVent precedent. Alliance Pharmaceutical pursued LiquiVent under an IND filed in the early 1990s, conducted phase I-II and phase III trials over approximately ten years, and discontinued the program in 2001 after total program expenditures that, while not separately disclosed, are believed to have approached or exceeded USD 200 million. The OriGen Biomedical 2013 acquisition of the LiquiVent dataset suggests a residual value in the safety package. For a non-therapeutic enhancement use such as high-G tolerance, the regulatory framework is unsettled, and the investor risk profile is dominated by the absence of an established indication, making conventional venture-capital investment unlikely without a parallel therapeutic indication (such as cardiac arrest neuroprotection, which Orixha is pursuing).
5.4 Dual-Use Technology Valuation
A central question for both defense planners and investors is whether validation of PFC ventilation in one domain reduces the cost of validation in others. The available evidence suggests partial but limited transfer. A successful Orixha-style cardiac arrest neuroprotection approval would establish manufacturing scale, pharmacovigilance infrastructure, and a clinical operator base that could in principle support military investigational use under 10 USC 1107 frameworks; however, the operational requirements for a deployable high-G or saturation diving system differ substantially from those of a hospital-based hypothermia device, and the engineering pathway from one to the other is non-trivial. The strongest dual-use linkage is in the underlying perfluorocarbon manufacturing supply chain, which is sensitive to per- and polyfluoroalkyl substances (PFAS) regulatory developments globally.
6. Regulatory Landscape
6.1 Medical Device and Drug Approval Pathways
Within the United States, a PFC respiratory liquid would be regulated as a drug or as a combination product under FDA jurisdiction, requiring an IND for clinical investigation and ultimately a New Drug Application (NDA) or Biologics License Application for marketing authorization; the associated liquid ventilator would be regulated as a medical device, most likely Class III under premarket approval requirements. Within the European Union, the equivalent pathway under the European Medicines Agency (EMA) and the Medical Device Regulation (Regulation (EU) 2017/745) would similarly require clinical evaluation and CE marking. The LiquiVent IND precedent provides a documented procedural template through phase III, although the program ultimately did not result in approval [4][6]. Humanitarian Device Exemption pathways exist for rare-condition indications but are not directly applicable to enhancement use cases.
6.2 Military Exemptions and Investigational Use Frameworks
10 U.S.C. § 1107 governs the administration of investigational new drugs to members of the U.S. Armed Forces and requires informed consent except where the President, on the recommendation of the Secretary of Defense and the FDA, waives that requirement on grounds of military operational necessity [23]. The waiver provisions in 21 CFR 50.23(d) and Executive Order 13139 set the procedural standards for such waivers, which have been used historically for vaccine and prophylactic countermeasure deployments. For an enhancement use such as liquid breathing, the regulatory question is whether a Force Health Protection rationale can be constructed within the existing waiver framework, which is unsettled and would likely require legislative or executive clarification. NATO partner nations operate under analogous but not identical investigational-use rules, and any allied program would require coordinated approval procedures.
6.3 Occupational Safety and Diving Regulations
Commercial saturation diving in U.S. waters is regulated under OSHA 29 CFR 1910 Subpart T (commercial diving operations) and U.S. Coast Guard 46 CFR Subchapter V. Internationally, the IMO Code of Safety for Diving Systems (Resolution A.831(19)) and IMO MSC.1/Circ.1547 provide the basic framework, supplemented by the IMCA D 014 international code of practice and the DMAC medical guidance documents [24][25]. The UK Health and Safety Executive (HSE) Diving at Work Regulations 1997 (S.I. 1997/2776) and the offshore-specific Approved Code of Practice L103 govern UK-sector operations, while Norwegian operations fall under the Petroleum Safety Authority (PSA, now Havtil) framework, and Brazilian operations under Agência Nacional do Petróleo (ANP) and NORMAM regulation. Any liquid-breathing diving operation would require explicit modification of these frameworks, which presently assume gas-phase respiration.
6.4 Ethical and Consent Considerations
The Belmont Report (1979) and the World Medical Association Declaration of Helsinki provide the foundational principles of human-subjects research ethics. Institutional Review Board (IRB) oversight under 45 CFR 46 in the United States and the equivalent Research Ethics Committee structures in EU member states impose informed-consent and risk-benefit balance requirements that are particularly searching for non-therapeutic, enhancement-oriented research in healthy military or civilian volunteers. The tension between operational necessity and the autonomy protective core of biomedical research ethics is well-documented in the military medicine literature and would be acute for a liquid-breathing trial in healthy subjects. The current literature does not establish a consensus framework for resolving this tension in enhancement contexts.
7. Geopolitical and Strategic Dimensions
7.1 Strategic Value of Extended Human Performance Envelopes
The strategic value of an extended human acceleration envelope or an extended saturation diving depth envelope depends on the specific operational scenario. Sustained acceleration tolerance beyond +9 Gz could in principle enable airframes designed for higher manoeuvre limits, although current generation fighter airframe design (F-35, J-20, Su-57) is already constrained more by structural and engine considerations than by pilot physiology, and unmanned platforms with no human-physiology constraint are progressively assuming high manoeuvre roles. Extreme-depth diving capability would have strategic value in undersea infrastructure inspection, covert seabed operations, mine countermeasures at depths beyond conventional ROV ranges, and submarine rescue. These remain hedged claims; the realized strategic value would depend on co-evolution of platforms, countermeasures, and adversary capabilities.
7.2 Comparative National Programs
Open-source reporting on Russian and Chinese liquid-breathing or extreme-depth human performance programs is sparse and frequently speculative. Russian state media in 2017 referenced an ostensible Foundation for Advanced Research Projects ("Russian DARPA") demonstration of dachshund liquid breathing in connection with submarine escape research; the demonstration was widely covered but has not been followed by peer-reviewed publication and should be treated as a single open-source data point with intelligence-assessment uncertainty. China's deep-sea capabilities have advanced substantially in manned submersibles (Jiaolong, Fendouzhe) and in cable-cutting and seabed-intervention tools, with a 2025 Mechanical Engineering report on a deep-sea cable-cutting device drawing strategic attention; whether this work extends to human-physiology liquid-breathing research is not established in the open record. Strict epistemic hedging is warranted: the absence of open-source evidence is not evidence of absence, but neither does it support assertive claims about adversary program scope.
7.3 Alliance and Technology Sharing Implications
The NATO Science and Technology Organization, particularly the Human Factors and Medicine Panel (HFM) and the Centre for Maritime Research and Experimentation (CMRE) at La Spezia, would be the principal multilateral channel for collaborative liquid-breathing research within the alliance [26]. The AUKUS Pillar 2 advanced capabilities framework, established under the September 2021 trilateral agreement among Australia, the United Kingdom, and the United States, includes an undersea capabilities working group whose principal current focus is on autonomous underwater vehicles (AUKUS Undersea Robotics Autonomous Systems) but whose remit explicitly extends to "additional undersea capabilities" [28]. Five Eyes life-sciences cooperation under the Technical Cooperation Program (TTCP) HUM (Human Resources and Performance) Group provides another existing mechanism. None of these mechanisms currently identifies liquid breathing as a named program element, and any allied collaboration would require new bilateral or multilateral agreements with associated technology-transfer and export-control implications.
7.4 Undersea Strategic Competition
Undersea infrastructure has emerged as a domain of strategic competition, with substantial 2024-2026 reporting on cable-cutting incidents in the Baltic Sea and around Taiwan, and with NATO's establishment of a Critical Undersea Infrastructure Coordination Cell in February 2023. The military significance of extended-depth manned capability lies in scenarios where ROV operations are insufficient (for example, in tasks requiring fine manipulation, sensor placement at depths beyond rated ROV operating envelopes, or covert infrastructure inspection where acoustic ROV signatures are operationally unacceptable). Available data suggest that the specific operational scenarios where extended manned depth would be decisive are narrow but high consequence; the evidence does not support a broad claim that liquid-breathing-enabled diving would transform undersea strategic competition, but it would offer asymmetric capability in selected niches.
8. Risk Matrix
The following matrix presents structured risk assessment across four categories and three time horizons. Likelihood and impact are coded L (low), M (medium), H (high).
Table 1. PFC Liquid Breathing Risk Matrix
| Category | Short-term (1–3 yrs) | Medium-term (3–7 yrs) | Long-term (7+ yrs) |
|---|---|---|---|
| Technical | CO2 washout ceiling in TLV remains unsolved at adult metabolic rates (L=H, I=H); driver: viscosity-limited mass transfer | Liquid ventilator miniaturization for field deployment unproven (L=M, I=H); driver: pump and oxygenator engineering | Integrated immersion plus TLV system for high-G unvalidated in humans (L=M, I=H); driver: lack of large-animal centrifuge data |
| Regulatory | No FDA-approved PFC respiratory drug exists (L=H, I=H); driver: LiquiVent discontinuation precedent | Investigational use under 10 USC 1107 untested for enhancement (L=M, I=H); driver: unsettled waiver framework | Allied harmonization (EMA, MHRA, TGA, Health Canada) absent (L=M, I=M); driver: divergent national pathways |
| Financial | Private capital reluctant without therapeutic indication (L=H, I=H); driver: post-LiquiVent caution | Defense procurement requires demonstrated TRL 6 system (L=M, I=H); driver: no current program of record | Total program cost ($1–3B) without committed offtake (L=M, I=M); driver: small addressable market |
| Adoption | Operator (pilot, diver) acceptability untested (L=H, I=M); driver: psychological barrier and precedent | Training pipeline and transition protocols undefined (L=M, I=M); driver: lack of operational doctrine | Insurance and liability frameworks unclear (L=M, I=M); driver: novel human-factors risk profile |
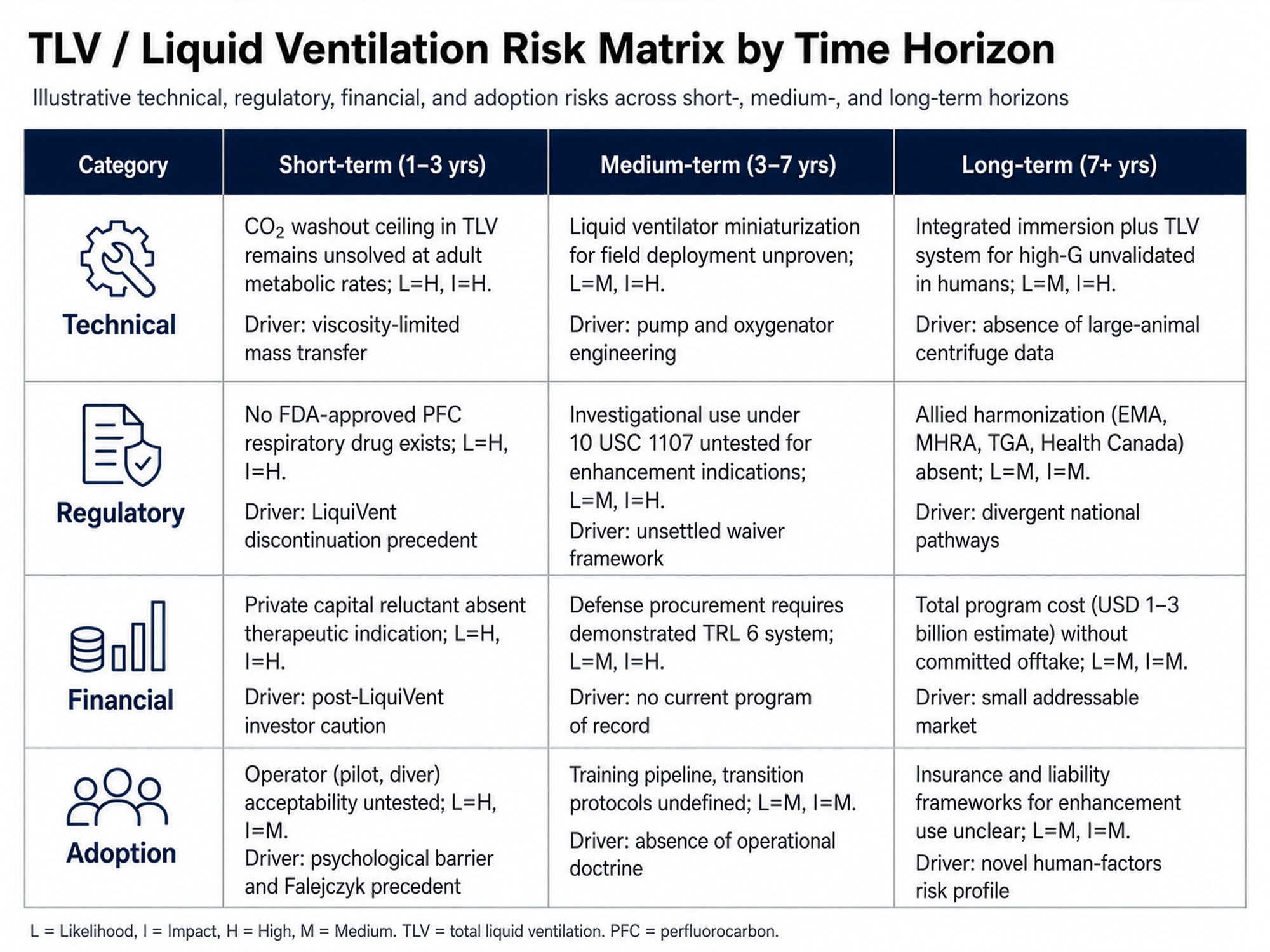
Interpretive analysis. The matrix shows compounding risk concentrations at the intersection of short-term technical and regulatory risk, where the unresolved CO₂ washout ceiling and the absence of an approved respiratory PFC create a foundational barrier that prevents medium term transitions in the financial and adoption categories from being meaningfully addressable. The dominant cluster, in both likelihood and impact terms, sits in the technical-regulatory short-term cell. This is the cell that any rational program plan must address first; investments in financial structuring or in adoption pathways before this cluster is resolved would be misallocated
A secondary cluster emerges in the medium term at the intersection of technical engineering risk (ventilator miniaturization and ruggedization) and financial risk (defense procurement requires technology readiness levels that current laboratory-grade Inolivent prototypes do not meet, and Orixha's clinical-hospital focus does not directly produce field-deployable hardware). Compounding here is significant because TRL advancement requires sustained funding that the absence of a procurement signal makes unlikely; this is a classic "valley of death" pattern familiar from the dual-use technology literature
The risk profile would shift materially under three identifiable conditions. First, an Orixha or successor clinical approval for cardiac-arrest neuroprotection would establish manufacturing scale, regulatory precedent, and a small but non-zero base of operator-physician training that would lower the marginal cost of subsequent investigational use. Second, a peer-reviewed large animal centrifuge experiment using TLV with acceleration tolerance as primary endpoint would either validate or falsify the core operational hypothesis; in the absence of such an experiment, the current debate is fundamentally inferential rather than evidentiary. Third, a clearly articulated adversary capability or strategic requirement (for example, a verified Russian or Chinese program achieving operational liquid-breathing capability in a defined niche) would change the political-economic calculus and create the procurement signal that the financial cluster requires.
The adoption-category risks are persistent across all time horizons but are individually of lower impact than the technical and regulatory categories. Operator acceptability concerns may be partially addressable through training analogues drawn from existing extreme-environment operator pipelines (saturation divers, astronaut candidates, test pilots) but would require dedicated psychometric study. Insurance and liability frameworks for enhancement-oriented use of investigational drugs in healthy operators are essentially undeveloped and would likely require legislative attention parallel to the technical program.
The matrix understates two correlated risks that warrant explicit identification. The first is reputational and ethical risk: any adverse event in early human trials, particularly one with a Falejczyk-style infectious or pulmonary morbidity, would have outsized consequences for the entire research line. The second is geopolitical-attribution risk: a successful enhancement capability achieved by one alliance bloc would create pressure on adversary blocs that may produce unsafe research practices and proliferation of incomplete protocols.
9. Strategic Recommendations
9.1 Audience A: Defense Policymakers and Military Program Offices
Recommendation A1. Commission a formal NATO Science and Technology Organization Human Factors and Medicine Panel research task group to assess the state of the art in PFC liquid ventilation as relevant to extended human performance envelopes, with explicit deliverables including a literature synthesis, a prototype-status assessment of Inolivent and Orixha hardware, and a NATO-coordinated research road map. This addresses the section 3.1 finding that allied research is fragmented and the section 7.3 finding that no current alliance mechanism explicitly covers this domain [26].
Recommendation A2. Authorize and fund a controlled large-animal centrifuge experiment combining total liquid ventilation with sustained +Gz exposure, with acceleration tolerance and post-exposure pulmonary recovery as primary endpoints, to be conducted at the 711th HPW centrifuge in collaboration with a TLV-capable academic partner. This addresses the section 4.2 finding that the core operational hypothesis is unvalidated and the section 8 risk-matrix dominant cluster.
Recommendation A3. Establish a memorandum of understanding among the 711th HPW, NEDU, NAMRU-Dayton, and a designated FDA liaison to clarify the regulatory pathway under 10 USC 1107 and 21 CFR 50.23 for non-therapeutic investigational use of PFC respiratory liquids in healthy military volunteers, and to draft a model IRB protocol that could serve as a precedent for subsequent trials [23]. This addresses the section 6.2 finding that the existing waiver framework is unsettled for enhancement applications.
Recommendation A4. Maintain the science under open peer review during the foundational physiology phase, with classification reserved for operational integration, ventilator hardware specifications, and any nation-specific tactical concepts of operation. Premature classification at the basic-physiology level would isolate the U.S. program from the Sherbrooke and Créteil collaborators who hold key engineering knowledge and would forgo allied scientific contributions [11].
Recommendation A5. Direct DARPA Biological Technologies Office and Office of Naval Research to issue a coordinated broad agency announcement targeting (a) CO₂ washout enhancement during TLV at adult metabolic rates and (b) ruggedized field-deployable liquid ventilator architecture, with explicit TRL transition milestones from 4 to 6 over a five-year horizon. This addresses the section 8 medium-term technical risk concentration [22].
Recommendation A6. Withhold any operational-readiness or fielding-decision authorization until peer-reviewed primate trials with sustained physiological exposure consistent with target operational profiles have been published. The Falejczyk precedent and the safety profile of perflubron at sub-acute exposure durations are insufficient bases for healthy-operator field deployment.
Recommendation A7. Through AUKUS Pillar 2 and Five Eyes life-sciences cooperation channels, initiate a discreet intelligence-community estimate on adversary research programs in liquid breathing and extreme human performance, recognizing the section 7.2 finding that current open-source reporting is sparse and frequently speculative [28].
9.2 Audience B: Institutional Investors and Deep-Sea Industry Executives
Recommendation B1. Treat any current commercial liquid-breathing investment proposition specifically targeting high-G or extreme-depth applications as occupying a technology readiness level no higher than TRL 3 to 4. The published large-animal evidence base supports a higher TRL only for cardiac-arrest hypothermia indications; investments justified by enhancement applications should be discounted accordingly. This reflects the section 4.6 finding that no controlled adult human TLV trials exist for these applications.
Recommendation B2. Concentrate near-term commercial diligence on Orixha and OriGen Biomedical as the principal entities with verifiable IP positions and clinical pathway artifacts, recognizing that both currently target indications adjacent to the high-G and extreme-depth use cases rather than those use cases directly [13]. Conditions for rational commercial investment include a successful Orixha cardiac-arrest phase II readout, a pre-NDA meeting with FDA producing favorable feedback, and demonstrated manufacturing scale-up of a perflubron or successor PFC compound
Recommendation B3. Structure any partnership with a defense-customer entity through an Other Transaction Authority arrangement or comparable allied mechanism rather than a conventional procurement contract, given the section 6.2 finding that regulatory uncertainty in enhancement use makes contract performance milestones difficult to specify in advance.
Recommendation B4. Conduct active intellectual property diligence focused on the Inolivent group patent portfolio (U.S. Patents 7,726,311 and related) and the Tissier-Berdeaux cooling with-liquid-ventilation patent (US20120226337A1), recognizing that any commercial product is highly likely to depend on cross-licensing with these holders [11][13]. The LiquiVent dataset acquired by OriGen represents a potentially valuable safety-data asset whose strategic positioning has not been fully realized.
Recommendation B5. Sequence regulatory engagement so that a clinical-therapeutic indication (cardiac arrest, ARDS, or a comparable critically ill population with established medical need) precedes any enhancement-oriented filing. The history of the LiquiVent program (section 5.3) demonstrates that even therapeutic indications have non-trivial completion risk; an enhancement-only filing would face compounded regulatory uncertainty.
Recommendation B6. For deep-sea industry executives, treat the addressable saturation diving market expansion enabled by liquid breathing as a tertiary investment thesis rather than a primary one, given the section 5.2 observation that current trends favor remotely operated and autonomous systems for tasks below approximately 300 msw. The strongest commercial case would arise from a defense-procurement-driven base load that brings a system to TRL 8 or 9 at government expense, after which spillover into commercial saturation diving could be evaluated.
Recommendation B7. Monitor PFAS regulatory developments in the European Union, the United States, and Japan as a potential category-level risk to the perfluorocarbon manufacturing supply chain, recognizing that perflubron and perfluorodecalin are subject to scrutiny under broader perfluoroalkyl regulatory initiatives even where their specific medical-grade applications are not the primary regulatory target.
10. References
[1] Clark, L. C., Jr., & Gollan, F. (1966). Survival of mammals breathing organic liquids equilibrated with oxygen at atmospheric pressure. Science, 152(3730), 1755–1756. https://doi.org/10.1126/science.152.3730.1755
[2] Kylstra, J. A. (1974). Liquid breathing. Undersea Biomedical Research, 1(3), 259–269.
[3] Wolfson, M. R., Greenspan, J. S., Deoras, K. S., Rubenstein, S. D., & Shaffer, T. H. (1992). Comparison of gas and liquid ventilation: Clinical, physiological, and histological correlates. Journal of Applied Physiology, 72(3), 1024–1031. https://doi.org/10.1152/jappl.1992.72.3.1024
[4] Leach, C. L., Greenspan, J. S., Rubenstein, S. D., Shaffer, T. H., Wolfson, M. R., Jackson, J. C., deLemos, R., & Fuhrman, B. P., for the LiquiVent Study Group. (1996). Partial liquid ventilation with perflubron in premature infants with severe respiratory distress syndrome. New England Journal of Medicine, 335(11), 761–767. https://doi.org/10.1056/NEJM199609123351101
[5] Wolfson, M. R., & Shaffer, T. H. (2005). Pulmonary applications of perfluorochemical liquids: Ventilation and beyond. Paediatric Respiratory Reviews, 6(2), 117–127. https://doi.org/10.1016/j.prrv.2005.03.010
[6] Hirschl, R. B., Croce, M., Gore, D., Wiedemann, H., Davis, K., Zwischenberger, J., & Bartlett, R. H., for the Adult Partial Liquid Ventilation Study Group. (2002). Prospective, randomized, controlled pilot study of partial liquid ventilation in adult acute respiratory distress syndrome. American Journal of Respiratory and Critical Care Medicine, 165(6), 781–787. https://doi.org/10.1164/ajrccm.165.6.2003052
[7] Hirschl, R. B., Conrad, S., Kaiser, R., Zwischenberger, J. B., Bartlett, R. H., Booth, F., & Cardenas, V. (1998). Partial liquid ventilation in adult patients with ARDS: A multicenter phase I–II trial. Annals of Surgery, 228(5), 692–700. https://doi.org/10.1097/00000658-199811000-00009
[8] Kacmarek, R. M., Wiedemann, H. P., Lavin, P. T., Wedel, M. K., Tütüncü, A. S., & Slutsky, A. S. (2006). Partial liquid ventilation in adult patients with acute respiratory distress syndrome. American Journal of Respiratory and Critical Care Medicine, 173(8), 882–889. https://doi.org/10.1164/rccm.200508-1196OC
[9] Riess, J. G. (2005). Understanding the fundamentals of perfluorocarbons and perfluorocarbon emulsions relevant to in vivo oxygen delivery. Artificial Cells, Blood Substitutes, and Immobilization Biotechnology, 33(1), 47–63. https://doi.org/10.1081/BIO-200046659
[10] Sekins, K. M., Wolfson, M. R., Stern, R. G., Greenspan, J. S., & Shaffer, T. H. (1994). Utility of a perfluorochemical liquid for pulmonary diagnostic imaging. Artificial Cells, Blood Substitutes, and Biotechnology, 22(4), 1409–1420. https://doi.org/10.3109/10731199409138845
[11] Robert, R., Micheau, P., Avoine, O., Beaudry, B., Beaulieu, A., & Walti, H. (2010). A regulator for pressure-controlled total-liquid ventilation. IEEE Transactions on Biomedical Engineering, 57(9), 2267–2276. https://doi.org/10.1109/TBME.2009.2031096
[12] Sage, M., You, W., Beaudry, É., Micheau, P., Walti, H., Fortin Pellerin, É., & Praud, J.-P. (2018). Perflubron distribution during transition from gas to total liquid ventilation. Frontiers in Physiology, 9, 1723. https://doi.org/10.3389/fphys.2018.01723
[13] Kohlhauer, M., Lidouren, F., Remy-Jouet, I., Mongardon, N., Adam, C., Bruneval, P., Hocini, H., et al. (2019). A new paradigm for lung-conservative total liquid ventilation. EBioMedicine, 52, 102365. https://doi.org/10.1016/j.ebiom.2019.08.026
[14] Bennett, P. B., & Rostain, J.-C. (2003). The high pressure nervous syndrome. In A. O. Brubakk & T. S. Neuman (Eds.), Bennett and Elliott's physiology and medicine of diving (5th ed., pp. 323–357). Saunders.
[15] Brauer, R. W., Dimov, S., Fructus, X., Gosset, A., & Naquet, R. (1969). Syndrome neurologique et électrographique des hautes pressions. Revue Neurologique (Paris), 121(3), 264–265.
[16] Lafay, V., Barthélémy, P., Comet, B., Frances, Y., & Jammes, Y. (1995). ECG changes during the experimental human dive HYDRA 10 (71 atm/7,200 kPa). Undersea and Hyperbaric Medicine, 22(1), 51–60.
[17] Jain, K. K. (1994). High-pressure neurological syndrome (HPNS). Acta Neurologica Scandinavica, 90(1), 45–50. https://doi.org/10.1111/j.1600-0404.1994.tb02678.x
[18] U.S. Navy. (2016). U.S. Navy diving manual, revision 7 (NAVSEA SS521-AG-PRO-010). Naval Sea Systems Command.
[19] Whinnery, J. E., & Forster, E. M. (2013). The +Gz-induced loss of consciousness curve. Extreme Physiology and Medicine, 2(1), 19. https://doi.org/10.1186/2046-7648-2-19
[20] Burton, R. R. (1988). Anti-G suit inflation rate requirements. Aviation, Space, and Environmental Medicine, 59(7), 601–605.
[21] Boissady, E., Kohlhauer, M., Lidouren, F., Hocini, H., Lefebvre, C., Chateau-Joubert, S., Mongardon, N., et al. (2020). Ultrafast hypothermia selectively mitigates the early humoral response after cardiac arrest. Journal of the American Heart Association, 9(23), e017413. https://doi.org/10.1161/JAHA.120.017413
[22] U.S. Defense Advanced Research Projects Agency. (2024). Broad agency announcement: Biological Technologies Office HR001124S0034. DARPA.
[23] U.S. Code. (2018). Title 10, Section 1107: Notice of use of an investigational new drug or a drug unapproved for its applied use. U.S. Government Publishing Office.
[24] International Marine Contractors Association. (2014). International code of practice for offshore diving (IMCA D 014 Rev. 2). IMCA.
[25] Diving Medical Advisory Committee. (2015). Medical equipment to be held at the site of an offshore diving operation (DMAC 15 Rev. 4). DMAC.
[26] NATO Science and Technology Organization. (2025). 2025 highlights report (NATO STO-004-2025). NATO STO.
[27] Imbert, J.-P., Matity, L., Massimelli, R. J., & Balestra, C. (2022). Review of excursion procedures used in commercial heliox saturation diving. Diving and Hyperbaric Medicine, 52(4), 277–290. https://doi.org/10.28920/dhm52.4.277-290
[28] Nicastro, L. A. (2024). AUKUS Pillar 2 (advanced capabilities): Background and issues for Congress (CRS Report R47599). Congressional Research Service.
[29] Tissier, R., Giraud, S., Quellard, N., Fernandez, B., Lidouren, F., Darbera, L., Chenoune, M., et al. (2014). Kidney protection by hypothermic total liquid ventilation after cardiac arrest in rabbits. Anesthesiology, 120(4), 861–869. https://doi.org/10.1097/ALN.0000000000000048
[30] Hutin, A., Lidouren, F., Kohlhauer, M., Lotteau, L., Seemann, A., Mongardon, N., Renaud, B., et al. (2015). Total liquid ventilation offers ultra-fast and whole-body cooling in large animals in physiological conditions and during cardiac arrest. Resuscitation, 93, 69–73. https://doi.org/10.1016/j.resuscitation.2015.05.029
[31] Greenspan, J. S., Wolfson, M. R., Rubenstein, S. D., & Shaffer, T. H. (1990). Liquid ventilation of human preterm neonates. Journal of Pediatrics, 117(1), 106–111. https://doi.org/10.1016/s0022-3476(05)82457-6
[32] Shaffer, T. H., Forman, D. L., & Wolfson, M. R. (1984). Physiological effects of ventilation with liquid fluorocarbon at controlled temperatures. Undersea Biomedical Research, 11(3), 287–298.
[33] U.S. Food and Drug Administration. (2018). Guidance for industry: Emergency use authorization of medical products and related authorities. FDA Office of the Commissioner.
[34] Centre for Strategic and International Studies. (2024). Safeguarding subsea cables: Protecting cyber infrastructure amid great power competition. CSIS.
[35] Lundgren, C. E. G., & Lin, Y. C. (1999). The lung at pressure: A historical perspective. Undersea and Hyperbaric Medicine, 26(Suppl.), 1–7.




